|
 |
Page At A Glance |
Table Of Contents |
Introduction
Background
SNOMED CT terminology provides a common language that enables a consistent way of indexing, storing, retrieving, and aggregating clinical data across specialties and sites of care.
SNOMED International maintains the SNOMED CT technical design, the content architecture, the SNOMED CT content (includes the concepts table, the descriptions table, the relationships table, a history table, and ICD mappings), and related technical documentation.
Purpose
This document provides a summarized description of the content changes included in the January 2021 release of SNOMED Clinical Terms® (SCT) International Release.
It also includes technical notes detailing the known content or technical issues where the root cause is understood, the fix has been discussed and agreed to, but has yet to be implemented.
The SNOMED International Release Notes are available alongside the January 2021 International Edition release.
Scope
This document is written for the purpose described above and is not intended to provide details of the technical specifications for SNOMED CT or encompass every change made during the release.
Audience
The audience includes National Release Centers, WHO-FIC release centers, vendors of electronic health records, terminology developers and managers who wish to have an understanding of changes that have been incorporated into the January 2021 International Edition release.
Content Development Activity
Summary
Continuous quality improvement and enhancement of existing content is an ongoing process undertaken by SNOMED International in preparation for every release. The January 2021 International Release has seen a continuation of the work driven by contributions from: Kaiser Permanente i.e. Convergent Medical Terminology (CMT), Global Medical Device Nomenclature Agency (GMDNA), Orphanet and other domain specific collaborations as well as requests received via the Content Request System (CRS).
Additionally quality improvement activities are advanced via project driven initiatives summarized below. Additional work items impacting every release are updates to the SNOMED CT derived maps such as ICD-10 and ICD-O; details are included in these release notes.
Information about editorial decisions may be found in the SNOMED CT Editorial Guide; mapping guidance for ICD-10 can be found at this link https://confluence.ihtsdotools.org/display/DOCICD10
The December 31st Member Release has been changed in time for the final Production Release on 31st January 2021. The Member Release should therefore be considered obsolete and should not be used for anything but test purposes. As always then, the final Production Release should be used in all systems. The reason for the changes was as communicated, in order to include important COVID-19 Vaccines content in the January 2021 International Edition, that wasn't available until January itself. Please see section 2.2 below for full details of these changes. In addition, four concepts were submitted via the CRS for inclusion into the January 2021 release accompanied with the incorrect concept ID. During testing it was identified the following new concepts had concept ID that did not match the descriptions in the National extension from which they originated: 28421000087109|Structure of right suprascapular region of back (body structure)| 28431000087106|Structure of left suprascapular region of back (body structure)| 28271000087107|History of peritoneal dialysis (situation)| 28281000087109|History of attention deficit hyperactivity disorder (situation)| The four invalid concepts along with all of their related components were "force deleted" from the SNOMED CT January 2021 International release after the Member release (31 December 2020) and before the Production release (January 2021). The four concepts will retain their current status in a National extension and be considered for promotion to the International release at a future date. Additional validation rules are being created to prevent future recurrence of this issue. |
| SCT Statistics | |
|---|---|
| Domain | #New concepts |
| SNOMED CT Concept (SNOMED RT+CTV3) | 3559 |
| Body structure (body structure) | 381 |
| Clinical finding (finding) | 1706 |
| Event (event) | 29 |
| Environment or geographical location (environment / location) | 0 |
| Observable entity (observable entity) | 180 |
| Organism (organism) | 164 |
| Pharmaceutical / biologic product (product) | 486 |
| Physical force (physical force) | 1 |
| Physical object (physical object) | 47 |
| Procedure (procedure) | 237 |
| Qualifier value (qualifier value) | 41 |
| Record artifact (record artifact) | 5 |
| Situation with explicit context (situation) | 108 |
| Social context (social concept)| | 6 |
| Specimen (specimen) | 16 |
| Staging and scales (staging scale) | 12 |
| Substance (substance) | 103 |
| SNOMED CT Model Component (metadata) | 37 |
| SCT Statistics for January 2021 Release | |
|---|---|
| #total | |
| Total number of active concepts | 354448 |
| Newly inactivated concepts | 3512 |
| Newly reactivated concepts | 17 |
| Sufficiently defined | 119603 |
| Primitive | 361906 |
COVID-19
Content relating to COVID-19 can be viewed here SNOMED CT COVID-19 Related Content
New content for January 2021 International release - Member Release |
|---|
1119305005 |Vaccine product containing only Severe acute respiratory syndrome coronavirus 2 antigen (medicinal product)| 1119349007 |Vaccine product containing only Severe acute respiratory syndrome coronavirus 2 messenger ribonucleic acid (medicinal product)| 1119350007 |Administration of vaccine product containing only Severe acute respiratory syndrome coronavirus 2 messenger ribonucleic acid (procedure)| |
Updated modeling and terming to conform with Editorial Guide for the following existing concept: 840534001 |Administration of vaccine product containing only Severe acute respiratory syndrome coronavirus 2 antigen (procedure)| Inactivated the following existing concept: 90640007 |Coronavirus vaccination (procedure)| |
To address the need for concepts related to persistent symptoms of COVID-19, three new concepts have been added to SNOMED CT for the January 2021 release. 1119302008 |Acute disease caused by Severe acute respiratory syndrome coronavirus 2 (disorder)| 1119303003 |Post-acute COVID-19 (disorder)| 1119304009 |Chronic post-COVID-19 syndrome (disorder)| |
897034005 Severe acute respiratory syndrome coronavirus 2 antibody test positive (finding) 897035006 Severe acute respiratory syndrome coronavirus 2 antibody test negative (finding) |
| 897036007 Occupational exposure to Severe acute respiratory syndrome coronavirus 2 (event) |
1017214008 Viremia caused by Severe acute respiratory syndrome coronavirus 2 (disorder)| |
Any concepts in scope for the SNOMED CT to ICD-10 mapping have been mapped and adhere to the World Health Organization current guidelines.
As part of the global effort to tackle the Coronavirus pandemic, SNOMED International has received several requests for content to be added for new vaccines as these have received approval by regulators in multiple countries. Although the requests were received after the new content deadlines for the January 2021 Release, new concepts have been added in consultation with requestors. These new concepts are deemed to be of low impact and low risk to the release process whilst being of extremely high importance to users of SNOMED CT. As vaccine-related requests and refinements to the original requests have been submitted to SNOMED International early in January 2021 this content has not been added to the 31st December 2020 Member Release, but instead to the final reviewed content in the Production Release on 31st January 2021. Further information including details about the new concepts is available here SNOMED CT COVID-19 Related Content |
Body structure
New body structure concepts: 381
Demonstration Release of the Anatomy Model
The plan is to implement the new anatomy concept model over forthcoming releases. There are approximately 35,000 anatomy concepts to be modeled by different types of 'part of' relationships. The new model will enable the automatic generation of hierarchies to further improve content quality and consistency. The integumentary system (about 2000 concepts) has been modeled. The musculoskeletal system (about 10,000 concepts) is the next hierarchy to be modeled. The concept model requires tooling enhancement to support nested expressions and further discussion about the inferred relationships for transitive and reflexive attributes. We will inform the community of the schedule for the demo release when the tooling and content are ready. The demo release will help us to gather feedback to evaluate potential impact and options for future releases.
Limbs/Girdles
There is mixed-use of the pelvic girdle and pelvic ring, shoulder girdle and shoulder in relation to the free limbs, limbs, and trunk. Existing content has been reviewed with about 200 model changes and impact to over 2000 classification results.
The following is a high-level view of hierarchical relationships for limb structures:
53120007 |Upper limb structure|
896766000 |Structure of free upper limb|
16982005 |Shoulder region structure|
26444007 |Shoulder girdle structure|
61685007 |Lower limb structure|
46862004 |Buttock structure|
29836001 |Hip region structure|
699617006 |Structure of free lower limb|
897012008 |Structure of bone and/or joint of pelvic girdle| - Note, this relationship is missing in the January 2021 release. It will be added in the next release.
The hierarchical relationships for trunk structures:
816092008 |Structure of pelvic cross-sectional segment of trunk|
46862004 |Buttock structure|
29836001 |Hip region structure|
12921003 |Structure of pelvis|
3665003 |Pelvic wall structure|
897011001 |Structure of pelvic ring|
897012008 |Structure of bone and/or joint of pelvic girdle|
Further detail can be found in the document https://docs.google.com/document/d/1zlVNTxuiL7UrvB8mjvDER0qJHzZjqe_ieVVfeqthNvc/edit?usp=sharing
Flexor annular pulley
Over 40 new concepts for flexor annular pulley of digits of hand have been added in this release.
Muscle tendon of toes
Over 20 new concepts have been added for flexor/extensor digitorum brevis/digitorum longus muscle tendons of toes.
Extensor compartment of wrist region
12 new concepts have been added for six extensor compartments of wrist region.
Nasal sinus structure
Update to the subhiearchy to clarify that sinus structure includes bone, cavity, and mucous membrane of sinus. New concepts have been added for bones and cavities of most nasal sinuses.
Lumbar region
The concept |Lumbar region| has been inactivated with reason ambiguous and has been replaced by new concepts |Lumbar region of abdomen| and |Lumbar region of back|.
Existing concepts that reference lumbar region, for example in the clinical finding and procedure hierarchy, have been reviewed and remodeled.
Interdigital webs and spaces of hand and foot
New concepts have been added for interdigital web of hand and interdigital web of foot and the subtypes have been assigned.
Cell hierarchy
The planned revision of Cell hierarchy has been put on hold. Further investigation is needed to determine if the SEP model should be applied for cell structure.
Anatomy changes to support Cancer Synoptic Reporting
New concepts have been added to support this project:
- Structure of lower thoracic esophagus (body structure)
- Structure of middle thoracic esophagus (body structure)
- Structure of upper thoracic esophagus (body structure)
Joint implant/articular surface
Improvements made for articular surface of limb joint body structure concepts so these will now enable proper classification of the joint implants while modeling content in the devices hierarchy.
Inactivation of Tooth Anatomy Concepts
On the recommendation of the Dentistry Clinical Reference Group, the following concepts have been inactivated due to their potential to pose a clinical risk:
| 39844006 |Structure of mandibular left canine tooth (body structure) | | 40005008 |Structure of mandibular right second molar tooth (body structure) | |
| 113278005 |Structure of mandibular left central incisor tooth (body structure) | | 72876007 |Structure of maxillary left canine tooth (body structure) | |
| 89625000 |Structure of mandibular left first molar tooth (body structure) | | 31982000 |Structure of maxillary left central incisor tooth (body structure) | |
| 77130001 |Structure of mandibular left lateral incisor tooth (body structure) | | 23427002 |Structure of maxillary left first molar tooth (body structure) | |
| 48402004 |Structure of mandibular left second molar tooth (body structure) | | 25748002 |Structure of maxillary left lateral incisor tooth (body structure) | |
| 47055002 |Structure of mandibular right canine tooth (body structure) | | 66303006 |Structure of maxillary left second molar tooth (body structure) | |
| 15422005 |Structure of mandibular right central incisor tooth (body structure) | | 80647007 |Structure of maxillary right canine tooth (body structure) | |
| 28480000 |Structure of mandibular right first molar tooth (body structure) | | 22120004 |Structure of maxillary right central incisor tooth (body structure) | |
| 82628004 |Structure of mandibular right lateral incisor tooth (body structure) | | 5140004 |Structure of maxillary right first molar tooth (body structure) | |
| 11712009 |Structure of maxillary right lateral incisor tooth (body structure) | | 7121006 |Structure of maxillary right second molar tooth (body structure) | |
The concepts have been inactivated with reason 'ambiguous' and a historical association target of the permanent and deciduous tooth. For example: Structure of mandibular left lateral incisor tooth (body structure) has been inactivated as ambiguous and replaced by Structure of deciduous mandibular left lateral incisor tooth (body structure) and Structure of permanent mandibular left lateral incisor tooth (body structure).
Reference set updates
Updated and validated release file for the lateralizable body structure reference set.
Updated and validated release files for the SEP refsets.
Clinical Finding
New concepts added for clinical finding hierarchy: 1702
Quality Initiative
The Quality Initiative (QI) project is the implementation of the Quality Strategy. After a successful pilot project for the July 2018 release the next stage has been implemented for subsequent releases including January 2021.
Quality improvement tasks were deployed to improve internal structural consistency and ensure compliance with editorial policy related to the stated modeling of content. Additionally, correction or addition of defining relationships was carried out to accurately reflect current clinical knowledge and ensure the semantic reliability of descriptions associated with a concept.
Total count of changes for the QI project:
- Stated: A total of 9463 concepts had changes made to the Stated relationships in the model.
- Inferred: A total of 15750 concepts affected by inferred changes.
| QI Project focus for January 2021 release (work has begun or been completed) | |
|---|---|
| 419199007|Allergy to substance (finding)| | 56246009|Hypertrophy (morphologic abnormality)| |
| 37782003|Damage (morphologic abnormality)| | 708039003|Inflammatory lesion (morphologic abnormality)| |
| 5294002 |Developmental disorder (disorder)| (excluding congenital disorders) | 4147007|Mass (morphologic abnormality)| |
| 87642003|Dislocation (morphologic abnormality)| | 708041002|Mechanical lesion (morphologic abnormality)| |
| 25723000|Dysplasia (morphologic abnormality)| | 52988006|Lesion (morphologic abnormality)| |
| 1806006|Eruption (morphologic abnormality)| | 36191001|Perforation (morphologic abnormality)| |
| 107682006|Fibrosis AND/OR repair abnormality (morphologic abnormality)| | 30217000|Proliferation (morphologic abnormality)| |
| 31883006|Fused structure (morphologic abnormality)| | 125671007|Rupture (morphologic abnormality)| |
| 76197007|Hyperplasia (morphologic abnormality)| | 19130008|Traumatic abnormality (morphologic abnormality)| |
Fracture of vertebra without spinal cord lesion (disorder)
Based on input from the Editorial Advisory Group and a member stakeholder:
269061001 |Fracture of vertebra without spinal cord lesion (disorder)| and descendants have been inactivated with a status of 'Ambiguous' and the historical association POSSIBLY EQUIVALENT TO = 50448004|Fracture of vertebral column (disorder)|. These concepts originated from ICD-9 to represent the notion of "Fracture of X without mention of Y".
This type of term is not represented in ICD-10 or ICD-11. As "without mention" is not semantically equivalent to "without" and could mean that the information was not available in the record abstraction, these concepts are considered to be ambiguous in their meaning.
Malignant neoplasm
Updates for content relating to malignant neoplasm have now been completed with the exception of those concepts which belong to the hierarchy 269475001 |Malignant tumor of lymphoid, hemopoietic AND/OR related tissue (disorder)|.
Allergy to vaccine
Editorial guidelines and content for concept pattern "Allergy to X vaccine" have been updated for the January 2021 Release.
Adverse reaction to vaccine
Editorial guidelines and content for concept pattern "Adverse reaction to X vaccine" have been updated for the January 2021 Release.
Iatrogenic disorder
Subtypes of 12456005 |Iatrogenic disorder (disorder)| that do not have the word iatrogenic in the FSN have been remodeled so that they are no longer a subtype of this concept.
New subtypes have been added to 12456005 |Iatrogenic disorder (disorder)| where the disorder specifies iatrogenic in the FSN.
Lichen sclerosus et atrophicus
25674000 Lichen sclerosus et atrophicus (disorder) has been inactivated with reason outdated and replaced with new concept 895454001 Lichen sclerosus (disorder).
A new morphology concept 895217008 Lichen sclerosus (morphologic abnormality) has been created and used to remodel the new concept and its 20 descendants. This improvement has resulted in 895454001 Lichen sclerosus (disorder) having no stated subtypes (with all Sufficiently Defined where possible).
Contact dermatitis
Updated the modeling for 40275004 |Contact dermatitis (disorder)| and subtypes to adhere with a newly constructed template.
Hernia of abdominal cavity
The finding site value for hernia disorder concepts (excluding obstructed hernias which are being managed separately) has been reviewed and changes made where required to improve classification results.
Fetal finding
The value for the 363698007 |Finding site| attribute for fetal concepts has been updated to << 123037004 |Body structure| MINUS << 55460000 |Fetal structure| unless the site is a unique body structure present during the fetal period.
The fetal semantics are captured using the 246454002 |Occurrence|=303112003 |Fetal period| relationship. These modeling changes have been applied to the fetal content within the Clinical finding hierarchy. Fetal procedure concepts will be reviewed as part of the Procedure QI work.
Fetal or neonatal effect of maternal medical problem
206002004|Fetal or neonatal effect of maternal medical problem (disorder)| and descendants have been inactivated. Replacement concepts that do not include 'or' in the descriptions have been added as subtypes of 70591005|Fetal disorder (disorder)| and 22925008|Neonatal disorder (disorder)|.
Spina bifida
67531005|Spina bifida (disorder) and subtypes have been remodeled and marked as sufficiently defined where appropriate.
'On Examination' and 'Complaining of' concepts
'On examination' and 'Complaining of' concepts have been inactivated in the International release and moved back to the UK extension.
Disorder X without Disorder Y
The vast majority of existing X without Y concepts originated from ICD-9 with the specific meaning of "X disorder without mention of Y disorder". As the phraseology indicates a lack of data about disorder Y as opposed to a specific exclusion, this type of concept has not been included in ICD-10, nor proposed for ICD-11, except in the case of "Traumatic brain injury without open intracranial wound".
Addition of new X without Y concepts may only be made under the following conditions:
- The request for new content must be accompanied by a rationale as to the difference between "X disorder without Y disorder" and "X disorder."
- Approval for addition is given by the Head of Terminology.
For the most part, existing X without Y concepts will be inactivated as AMBIGUOUS with a historical MAY BE relationship to "X disorder". Exceptions to inactivation will be made on a case-by-case basis.
Ventricular outflow tract obstruction
Occurrence 'congenital' has been removed from 253546004|Left ventricular outflow tract obstruction (disorder) and 253530007 |Right ventricular outflow tract obstruction (disorder) with remodel of subtypes.
Human immunodeficiency virus
Human immunodeficiency virus Centers for Disease Control and Prevention and Human immunodeficiency virus World Health Organization class content has been updated to align with the current categories.
Chromosome replaced with ring or dicentric
268294001 |Chromosome replaced with ring or dicentric (disorder)| has been inactivated and replaced by 1010276004 |Ring chromosome (disorder). All subtypes have been remodeled and marked sufficiently defined where appropriate. 23345003 |Ring chromosome (morphologic abnormality)| has been remodeled to align with expert input to have monosomy, trisomy and ring chromosome concepts as siblings in the morphology hierarchy.
Review of 419099009 |Dead (finding)| and 397709008 |Patient died (finding)|
397709008 |Patient died (finding)| inactivated as a "Duplicate component" with historical association to 419099009 |Dead (finding)|. All of the subtypes of 397709008 |Patient died (finding)|moved to hierarchy 366044004 |Finding of place of death (finding)| concepts that include 'patient' in FSN inactivated with status outdated and replaced by concept that does not use 'patient' in the descriptions. 304215001 |Patient died during operation (finding)| inactivated and replaced with 876883006 |Died during operation (finding)| subtype of 183676005 |Died in hospital (finding)|. 184297005 |Patient died in hospital (finding)| inactivated as a duplicate of 183676005 |Died in hospital (finding)|
Nutrition finding
Remodeled relationships of descendants of 366358006 |Finding of nutrient intake (finding)| to align with template Nutrition intake (finding) - v1.0 (Please note, you may have to register for Confluence user account in order to access this link).
Approximately 160 new definitions for nutrition content promoted from the US Extension with the approval of the Nutrition Clinical Reference Group.
Social Determinants of Health (SDoH) - Clinical Finding
In line with SNOMED CT priorities, and working with the US Extension of SNOMED CT to make Social Determinants of Health (SDoH) content available for international usage on the subject matter of food insecurity. This work was based on submissions from the US CRS for content for SDoH originating from the consensus-driven standards being developed by the Gravity project: https://confluence.hl7.org/display/GRAV/The+Gravity+Project
A new concept was added with a definition: 1078229009 |Food security (finding)|
New definition for 733423003 |Food insecurity (finding)|
Also supporting SDoH work:
1010682008 |Not entitled to social services assistance due to income above limit (finding)|
Content Tracker Project Updates
Work completed for the following Content Project:
- IHTSDO-668 Congenital absence vs. congenital partial absence.
Notice: Inactivation reason of LIMITED/WAS_A is not allowed for any new content inactivations after the July 2018 release. The WAS_A association refset has not been updated thereafter. At the Editorial Advisory Group meeting in April 2019, agreement was reached to discontinue the maintenance of WAS_A relationships when inactivating concepts that have a historical association to an inactive concept. When changes are made to a historical relationship for a concept that was previously inactivated using WAS_A, effort will be made to assign a new historical relationship that facilitates traceability of the concept (e.g. DUPLICATE or AMBIGUOUS) as opposed to NON-CONFORMANCE TO EDITORIAL POLICY. Existing WAS_A relationships will be inactivated in a future release once a plan for batch reassignment of historical relationships has been developed. Until then, SNOMED International will not continue to use or maintain WAS_A relationships. |
|---|
Notice: 'Co-occurrent and due to' pattern: During the implementation of the new Description Logic features, a conflict was uncovered between the modeling of 'Co-occurrent and due to' and General Concept Inclusions (GCIs). This has resulted in the need to reconsider the modeling of "Co-occurrent and due to' and update the Editorial Guide for this area. The Editorial Guide and all concepts that are currently modeled as 'Co-occurrent and due to' will be updated over future release cycles. |
|---|
Convergent Medical Terminology (CMT)
New CMT concepts: 621
Procedure
New concepts for procedure hierarchy: 237
Computed tomography angiography
After consultation with clinical stakeholders and the Editorial Advisory Group, the following changes have been implemented:
- 418272005|Computed tomography angiography (procedure)| and all subtypes have been remodeled to include attribute 424361007 |Using substance (attribute)| and value 385420005 |Contrast media (substance)| in the role group.
- The descriptions for 418272005|Computed tomography angiography (procedure) and subtypes have been updated to explicitly state ‘with contrast.’
- Where logical equivalence has been identified once the above changes have been implemented one of the concepts has been inactivated, in these cases the existing concept that stipulates 'with contrast' in the FSN has been retained.
Fluoroscopic angiography
After consultation with clinical stakeholders and the Editorial Advisory Group, the following changes have been implemented:
- Remodeling of 420040002|Fluoroscopic angiography (procedure)| and all subtypes to include attribute 424361007 |Using substance (attribute)| and value 385420005 |Contrast media (substance)| in the role group.
- The descriptions for 420040002|Fluoroscopic angiography (procedure)| and subtypes have been updated to explicitly state ‘with contrast.'
- Remodeling as described above for 743 concepts.
- New descriptions were added to 640 concepts, including Fully Specified Name, Preferred Term and all synonyms.
- 2 new concepts were added.
- 117 Primitive concepts were made Sufficiently Defined - Note where logical equivalence was identified once the above changes were implemented this resulted in 11 concepts being inactivated, in these cases the existing concept that stipulated contrast in the FSN was retained.
Administration of vaccine
Updated inactivation reason to Outdated for "administration of vaccine products" that refer to "double" or "triple" but not for those that also refer to additional vaccines.
Social Determinants of Health (SDoH) Procedure
New evaluation procedure concepts:
1004109000 |Assessment of goals to achieve food security (procedure)|
1002223009 |Assessment of progress toward goals to achieve food security (procedure)|
1002224003 |Assessment for food insecurity (procedure)|
1002225002 |Assessment of barriers in food insecurity care plan (procedure)|
Also
1004110005 |Coordination of resources to address food insecurity (procedure)|
Laboratory procedure
Inactivation of 3 misplaced laboratory procedure concepts:
167350008 |Urine microscopy: no cells (procedure)|
167351007 |Urine microscopy: no epithelial cells (procedure)|
167352000 |Urine microscopy: no white cells (procedure)|
All referred to new findings concepts.
Nutrition procedure
Added one procedure concept for Nutrition: Administration of parenteral nutrition (procedure)|.
Remodeled relationships of many descendants of 182922004 |Dietary regime (regime/therapy)| to align with template.
Content Tracker Project Updates
Work continues on content tracker IHTSDO-175.
There are ongoing structural changes for the Procedure concepts related to the content tracker. Work on this project will continue for future releases after July 2021.
Collaboration/Harmonization Agreements
Orphanet
Working in collaboration with Orphanet (http://www.orpha.net/consor/cgi-bin/index.php), creation of new concepts for the original set of prioritized rare diseases has been completed.
The alpha release is now available for feedback, for more information please see April 2020 SNOMED CT Orphanet Simple Map package ALPHA release available
All of the concepts added for the Orphanet project have been mapped to ICD-10.
ICD-11 Update
A total of 500 new concepts were added for the January 2021 release.
Global Medical Device Nomenclature Agency (GMDNA)
33 new device concepts were added to the physical object hierarchy for GMDN for the January 2021 release.
For the GMDN mapping equivalence tables:
- 33 new concepts have been added to in scope and mapped.
- 199 concepts have been modified and removed from the mapping tool as out of scope.
- 84 concepts have been deemed obsolete and removed from the mapping tool and flagged as out of scope.
- 16 concepts have been flagged as out of scope for SNOMED CT and removed from this map.
ICD-O-3.2 Update
6 new morphologies have been added to the 108369006 |Neoplasm (morphologic abnormality)| sub-hierarchy and mapped to ICD-O-3.1, or researched and mapped to 'no target' as a suitable morphology code is not available in ICD-O-3.1:
- 1078227006 |High-grade non-invasive papillary urothelial carcinoma (morphologic abnormality)|
- 128804002 |Primary cutaneous CD30 antigen positive T-cell lymphoproliferative disorder (morphologic abnormality)|
- 397353001 |Lymphomatoid papulosis (morphologic abnormality)|
- 878856000 |Malignant lymphomatoid granulomatosis (morphologic abnormality)|
- 878865007 |Rhabdomyomatous mesenchymal hamartoma (morphologic abnormality)|
- 878881002 |Trichoblastoma (morphologic abnormality)|
Note - The published maps to ICD-O-3.1 are being updated only for maintenance purposes for the January 2021 release. The maps to ICD-O-3.2 are out of scope for the January 2021 release, and are planned for a future release after the publication of the ICD-O-3.2 classification.
Event
New concepts added: 29
Qualifier Value
New concepts added: 41
Social context
New concepts added: 6
Inactivation of subtypes of 372148003|Ethnic group (ethnic group)| - concepts which include "other" or relate to UK census have been inactivated from the International release and moved back to the UK extension.
Situation with explicit context
New concepts added: 109
Content that included the word 'unilateral' in the FSN has been reviewed and inactivated with a status of ambiguous where appropriate. Replacement left and right concepts have been created in the clinical finding or procedure hierarchy where required.
Physical object
New concepts added: 47
Concepts inactivated: 10
- Approximately 420 "Has compositional material" attribute and value relationships were added to concepts in the physical object hierarchy, e.g., 415529004 |Silicone punctal plug (physical object)| was given a relationship of "Has compositional material (attribute)" with value of 13652007 |Silicone (substance)|.
- Approximately 100 attribute-value relationships were added to the physical object hierarchy for device characteristic of being coated or uncoated.
- Approximately 35 attribute-value relationships were added to the physical object hierarchy for device intended sites including head of femur and acetabulum.
- Approximately 50 gastrointestinal implant device concepts in the physical object hierarchy were modeled with the new device attributes. Some of these concepts had FSN and description changes as well.
Specimen
New concepts added: 16
Special concept
362955004 |Inactive concept (inactive concept)| and its seven descendants have been inactivated. When the first release format (RF1) was used, inactivated content was moved into this hierarchy. However, this approach is no longer used in the current release format (RF2).
Observable Entity
Added 180 new concepts, mainly in the area of nutritional intake such as Measured quantity of fat and oil intake in 24 hours (observable entity).
- Inactivated ~30 dietary concepts including "Dietary X intake (observable entity)" e.g., 162513004 |Dietary fat intake (observable entity)| and Prescribed dietary intake (regime/therapy)
- Removed the Scale type attribute and value relationship from ~200 nutritional intake observable entity concepts to align with current editorial policy.
- Updated the Property attribute value and FSN for ~100 nutritional intake observable entity concepts
Organism
New organism concepts added: 164
Organism name changes: 101
Pharmaceutical/Biological Product
For further details on the planned changes in this area, please refer to the Drug Model Working Group Directory section. Please note, you may have to register for Confluence user account in order to access this project and the relevant link |
Active immunity stimulant therapeutic role
318331000221102 |Active immunity stimulant therapeutic role (role)| updated based on feedback received and with agreement from Drugs WG:
New FSN = Active immunity stimulant role (role)
New PT = Active immunity stimulant role
Stated parent changed from 766941000 |Therapeutic role (role)| to 766940004 |Role (role)|
Biological agent (product)
High-level grouper 69509008 |Biological agent (product)| was inactivated based on agreement from Drugs Working Group; the concept did not have any stated or inferred descendants.
Vaccine product "containing" groupers
Vaccine product "containing" groupers that are modeled with more than one active ingredient or with an active ingredient that is a substance modification have been inactivated as out of scope per the editorial guidelines.
Blood derivative product (product)
High-level grouper 346348003 |Blood derivative product (product)| was inactivated based on agreement from Drugs Working Group; the descendants were reviewed and remodeled using the existing medicinal product concept model.
Has ingredient characteristic and Has product characteristic attribute
Based on feedback provided by the Editorial Advisory Group, the Has ingredient characteristic and Has product characteristic attributes will be refined to more specific attributes in a future release.
Medical gases
349362006 |Medical gases (product)| was inactivated as outdated; descendants were remodeled per existing Medicinal product editorial guidelines. Exceptions to allow 385217004 |Conventional release gas for inhalation (dose form)| as a manufactured dose form for Medicinal product form concepts and synonyms for percent and parts per million strengths for Clinical drug concepts added to editorial guidelines. Note that existing concepts such as 320917000 |Oxygen gas (product)| or 421396006 |Helium + oxygen gas (product)|were remodeled as MP-only concepts as per decision made earlier in Medicinal product project.
Roles
Modeling of roles for medicinal product concepts was initiated; a log of roles modeled is available at Drugs Project in anticipation of removing product role groupers from the Substance hierarchy and using Medicinal product concepts as attribute values in the International Release for modeling concepts in other SNOMED CT hierarchies.
TallMan lettering
The Drugs Working Group agreed TallMan Lettering should be addressed in a National Extension, not the International Release.
Veterinary Extension
2 concepts were moved to the Veterinary Extension.
Metadata concept
New concepts added: 37
New metadata concepts have been added to support postcoordinated expressions.
- Reference set concepts:
1119417006 |Postcoordinated expression type reference set (foundation metadata concept)|
1119435002 |Canonical close to user form expression reference set (foundation metadata concept)|
1119468009 |Classifiable form expression reference set (foundation metadata concept)|
- Reference set attribute concepts:
Subtypes of 706999006 |Expression (foundation metadata concept)|:
1119454002 |Compositional grammar expression (foundation metadata concept)|
1119455001 |Close to user form expression (foundation metadata concept)|
1119436001 |Canonical close to user form expression (foundation metadata concept)|
- Subtypes of 900000000000457003 |Reference set attribute (foundation metadata concept)|:
1119456000 |Substrate (foundation metadata concept)|
733616009 | Authoring substrate (foundation metadata concept) |
1119457009 |Classification substrate (foundation metadata concept)|
- Attribute type concepts:
Subtypes of 707000009 |SNOMED CT parsable string (foundation metadata concept)|:
1119437005 |SNOMED CT compositional grammar (foundation metadata concept)|
1119458004 |SNOMED CT expression constraint language (foundation metadata concept)|
1119459007 |SNOMED CT expression template language (foundation metadata concept)|
- Subtype of 900000000000460005 |Component type (foundation metadata concept)|:
1119460002 |Postcoordinated expression type component (foundation metadata concept)|
- Subtype of 900000000000460005 |String (foundation metadata concept)|:
1119461003 |Uniform resource identifier (foundation metadata concept)|
900000000000469006 | Uniform resource locator (foundation metadata concept)|
Substance
New concepts added: 103
New disposition and Disposition grouper concepts added: 6
As part of the Substance hierarchy maintenance, 81118006 |Gonadal hormone (substance)| was inactivated and replaced with 312263009 |Sex hormone (substance)|. The concepts previously subsumed by Gonadal hormone were remodeled where applicable.
For further details on the planned changes in this area, please refer to the Substances project. Please note, you may have to register for Confluence user account in order to access this project and the relevant links above. |
Internal Quality Improvement
Replacement of the Stated Relationship files with the new OWL Axiom refset files
A set of documentation has been developed to support the Logic Profile Enhancements.
- SNOMED DL Profile Enhancements
- SNOMED CT Logic Profile Specification
- SNOMED CT OWL Guide (OWL Refsets specification)
- Snomed OWL Toolkit - https://github.com/IHTSDO/snomed-owl-toolkit
- Classifying SNOMED CT using the Snomed OWL Toolkit - https://youtu.be/-91egY9mJqA
- Creating an OWL file containing SNOMED CT - https://youtu.be/sfFbMMioA_4
For any questions, please contact SNOMED International at support@snomed.org with “OWL Axiom refset files implementation question” in the subject line.
Machine Readable Concept Model (MRCM) Changes
Changes to the MRCM for the January 2021 release include:
- MRCM for new attribute |Unit of presentation size quantity|
- MRCM for new attribute |Unit of presentation size unit|
- MRCM: Expand the range of 370133003 |Specimen substance (attribute)| to include << 373873005 |Pharmaceutical / biologic product (product)| and << 260787004 |Physical object (physical object)|
- MRCM: Expand the range of 246501002 |Technique (attribute)| to include <<254291000 |Staging and scales (staging scale)
Further detail can be found at MRCM changes in the January 2021 release (Please note, you may have to register for Confluence user account in order to access this project and the relevant links above).
Concrete Domains and Numeric Representation
Please see here (and the various sub-pages) for details of the current proposed technical changes: https://confluence.ihtsdotools.org/mag/concrete-domains-community-of-practice-consultation. (Please note, you may have to register for Confluence user account in order to access this link).
In this child page in particular there is guidance on the impact for both maintainers of SNOMED CT plus for implementers and vendors of software that utilises the terminology: https://confluence.ihtsdotools.org/mag/concrete-domains-community-of-practice-consultation/snomed-international-proposal-for-representing-concrete-domains-in-rf2
Please, however, be aware that the technical proposal is still in the consultation stage, and therefore changes to the proposal may still be made before the final solution is implemented.
The transition has been discussed and agreed with numerous stakeholders from the community, and as such is targeted to be implemented over the course of the next two International Release cycles, as follows:
Q1 2021:
- A technical preview will be published in line with the January 2021 International Edition, with drug concept strengths and counts expressed as concrete values in the new Relationships file.
- This is currently planned to be a full RF2 International Edition package (as opposed to a standalone Delta file containing Concrete Domains), containing all International content in addition to the new Concrete Domains Relationship file
- (ie) it will comprise the January 2021 International Edition release as if we had already transitioned to Concrete Domains, as a preview of what the final Release package will look like when the transition is complete.
Q3 2021:
- The current target is for the July 2021 International Edition to be the first release after the transition to Concrete Domains. This means that the RF2 package would be impacted as follows:
- existing drug concept strength and count relationships inactivated +
- existing strength and count attribute types will be inactivated, replaced with new ones using the same FSNs.
- Inferred Relationship file changes:
- Stated OWL Axiom file changes:
- existing drug concept axioms will be updated to use concrete values +
- existing strength and count attribute types will be inactivated, replaced with new ones using the same FSNs.
- Additional Features in International Release
- new separate concrete value relationship file will express these same attributes using numeric values
- new attributes will be used, although they will have the same FSNs as the current attributes
- MRCM will include new rows to indicate that the new attribute types are expected to take a concrete domain - specifically numbers - as target values.
If you can foresee any critical issues with the transition plans, or have any questions regarding any of these upcoming changes, please contact SNOMED International immediately at support@snomed.org with “Concrete Domains transition question” in the subject line.
SNOMED CT derived products
ICD-10 map
The SNOMED CT to the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (© World Health Organization 1994) 2016 Version map (SNOMED CT to ICD-10 Map) is included in the SNOMED CT International release as a Baseline. The SNOMED CT to ICD-10 Map was created to support the epidemiological, statistical and administrative reporting needs of SNOMED International member countries and WHO Collaborating Centers.
The SNOMED CT to ICD-10 Map is released in Release Format 2 (RF2) only. It is located in the file der2_iisssccRefset_ExtendedMapFull_INT_20200731.txt, which is in the Map folder under Refset, in each of the three RF2 Release Type folders.
The SNOMED CT to ICD-10 Map is released as Refset 447562003 |ICD-10 complex map reference set (foundation metadata concept)|.
Content development activity summary
The map is a directed set of relationships from SNOMED CT source concepts to ICD-10 target classification codes. The SNOMED CT source domains for the MAP are limited to subtypes of 404684003 |clinical finding|, 272379006 |event| and 243796009 |situation with explicit context|. The target classification codes are ICD-10 2016 release.
Mapped content for January 2021
The map provided for the January 2021 release has been updated, and now represents a complete map from SNOMED CT International release to ICD-10 2016 version.
- 1873 newly authored concepts have been added and mapped
- 3 new codes for special purposes have been added to the terminology browser in accordance with WHO guidelines:
· U08 Personal history of COVID-19
· U09 Post COVID-19 condition
· U10 Multisystem inflammatory syndrome associated with COVID-19
We would welcome feedback on any issues that users of the map may detect when using the map. Issues should be submitted via mapping@snomed.org
Technical Guide Exemplars
The Technical Guide Exemplars document has now been moved from the International Edition release package to a Confluence page. This page can be found as part of the ICD-10 Mapping Technical Guide (see Appendix B), which is hosted here: http://snomed.org/icd10map
SNOMED CT to OWL conversion and classification
The repository containing the toolkit enabling simple SNOMED CT to OWL conversion and classification can be found here, including documentation on its use: https://github.com/IHTSDO/snomed-owl-toolkit
Please contact SNOMED International at support@snomed.org if you would like to provide any feedback on ways to extend and improve the new toolkit.
Technical notes
Known Issues
Known Issues are content or technical issues where the root cause is understood, and the resolution has been discussed and agreed but has yet to be implemented. This can be due to a number of reasons, from lack of capacity within the current editing cycle, to the risk of impact to the stability of SNOMED CT if the fix were to be deployed at that stage in the Product lifecycle.
For the current SNOMED CT International edition, the following Known Issues were identified, and agreed to be resolved in future editing cycles:
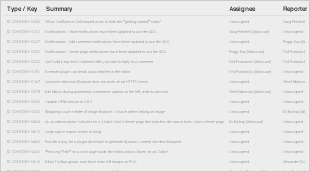
Resolved Issues
Resolved issues are Known Issues which were not fixed as part of the previous release lifecycle, but which have now been resolved in the latest release. They can also be issues found during the Alpha and Beta testing of the current release, which were resolved before the final deployment of the associated Member release. Finally they can be issues which were reported or found during the testing phase, but which have been closed without any action taken.
The Resolved Issues for the current Snomed CT International edition can be found here:

Technical updates
RF2 package format
For future reference, the RF2 package convention dictates that it contains all relevant files, regardless of whether or not there is content to be included in each particular release. Therefore, the package contains a mixture of files which contain both header rows and content data, and also files that are intentionally left blank (including only a header record). The reason that these files are not removed from the package is to draw a clear distinction between files that:
- have been deprecated (and therefore removed from the package completely), due to the content no longer being relevant to RF2 in this or future releases, and
- happen to contain no data in this particular release (and are therefore included in the package but left blank, with only a header record), but are still relevant to RF2, and could therefore potentially contain data in future releases.
This allows users to easily distinguish between files that have purposefully been removed or not, as otherwise if files in option 2 above were left out of the package it could be interpreted as an error, rather than an intentional lack of content in that release.
Configuration file in the RF2 package, containing Release Metadata
A new file has been included since the July 2020 International Edition, containing metadata about the Release package itself. This has been created in conjunction with feedback from the community, and as such initially contains the following fields:
- effectiveTime
- deltaFromDate
- deltaToDate
- languageRefset(s)
- humanReadableLanguageRefset(s)
- licenceStatement
The file is in .JSON format, to ensure that it is both machine-readable and human-readable, and is named "release_package_information.json".
The metadata will be extended and refined going forward, in order to ensure that it contains the most useful information possible. If you have any ideas about any other useful information to include, please send them to info@snomed.org, along with a business case explaining how the information would benefit stakeholders. Please be aware that this use case will then be assessed by SNOMED International, and the new metadata will only be included in the configuration file if the business case is strong enough.
New Refset Descriptor records
A reference set type has been created to give postcoordination implementations the option of storing expressions in RF2 format. A reference set of this type has been created for the "Canonical close to user form" and for the "Classifiable form" of expressions. Guidance on using these expressions will be provided during 2021.
Nine new records have therefore been added to the refsetDescriptor file for these new expression refsets. These do not correspond to actual refsets in the International Edition release package, they are being introduced in order to support the implementation of a postcoordinated expression repository:
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119417006 449608002 1119460002 0
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119468009 449608002 1119460002 0
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119468009 1119454002 1119437005 1
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119435002 1119436001 1119437005 1
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119417006 1119456000 1119461003 2
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119435002 733616009 1119461003 2
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119468009 1119457009 1119461003 2
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119417006 1119454002 1119437005 1
- (UUID) 20210131 1 900000000000012004 900000000000456007 1119435002 449608002 1119460002 0
Members Release now Obsolete
The December 31st Member Release has been changed in time for the final Production Release on 31st January 2021. The Member Release should therefore be considered obsolete and should not be used for anything but test purposes. As always then, the final Production Release should be used in all systems.
The reason for the changes was as communicated, in order to include important COVID-19 Vaccines content in the January 2021 International Edition, that wasn't available until January itself. Please see section 2.2 of these Release notes for full details of these changes.
In addition, four concepts were submitted via the CRS for inclusion into the January 2021 release accompanied with the incorrect concept ID. During testing it was identified the following new concepts had concept ID that did not match the descriptions in the National extension from which they originated. Please see section 2.1 of these Release notes for full details of these changes.
Concrete Domains Technical Preview
A technical preview of the impending Concrete Domains changes (currently planned for the July 2021 International Edition) will be published in February 2021, but in line with the January 2021 International Edition. It will contain drug concept strengths and counts expressed as concrete values in a new Relationships file. This is currently planned to be a full RF2 International Edition package (as opposed to a standalone Delta file containing Concrete Domains), containing all International content in addition to the new Concrete Domains Relationship file (ie) it will comprise the January 2021 International Edition release as if we had already transitioned to Concrete Domains, as a preview of what the final Release package will look like when the transition is complete.
Early visibility of impending change in the July 2021 International edition
Please see the following page for details of all upcoming changes planned for July 2021 and beyond: https://confluence.ihtsdotools.org/display/RMT/July+2021+Early+Visibility+Release+Notice+-+Planned+changes+to+upcoming+SNOMED+International+Release+packages
Document links
All links provide information that is correct and current at the time of this Release. Updated versions may be available at a later date, but if so these will need to be requested from the relevant SNOMED International teams.
NOTE: To access any of the links in the pdf document, please visit the Release Notes @ https://confluence.ihtsdotools.org/display/RMT/SNOMED+CT+January+2021+International+Edition+-+SNOMED+International+Release+notes |
Approvals
|
Draft Amendment History
|