Overview
Any SNOMED CT subset used to code COVID-19 data should be designed based on its purpose and the context in which it will be used.
To make the process of planning and designing your SNOMED CT subsets more efficient and internationally consistent, we recommend the following three steps.
These three steps are explained in more detail below.
Step 1 - Identify Data Elements
The first step is to identify the data elements that may require SNOMED CT coding. These data elements may already be defined in national reporting requirements 123, as part of a healthcare data standard you are using (e.g. a HL7 FHIR profile4 or openEHR archetypes5), or you may be developing a new information model (or data set) for this purpose. Your requirements for coded content will depend on your specific use case and the data items in your information model. In addition, different countries, regions, and hospitals may apply different clinical techniques or practices, which can also result in differing coding requirements. Therefore, deciding which subsets to implement requires clarification of the scope of content needed, by answering questions such as:
- What will your SNOMED CT subsets be used for?
- Will they be used to capture new data in a clinical information system to support frontline service delivery?
- Will they be used for disease surveillance?
- Will they be used to integrate data from various sources?
- Will they be used for international collaboration?
- Will they be used for retrospective analysis of data?
- Which data elements are required for your use case?
- What coded data needs to be collected to support clinical care?
- What coded data needs to be collected to provide disease surveillance?
- What coded data needs to be shared between or integrated from different sources?
- What coded data is required for international collaboration?
- What coded data may be needed for retrospective data analysis?
- What existing subsets are available for the data elements of interest?
- Where these subsets designed based on the same or compatible requirements?
Answering these questions will help in understanding which of the existing subsets are relevant for your context. It may also help to identify potential content gaps in the subsets, or extraneous concepts which are not required for your specific scenario. Please note that the questions above are provided for inspiration only. Additional questions specific to your use case are likely to be needed.
Step 2 - Download Subsets
The second step is to download any available existing subsets that are associated with your required data elements.
The example COVID-19 subsets in this guide can be downloaded from Appendix A - Example Subsets.
Subset Naming Convention
The example COVID-19 subsets provided by SNOMED International have been named using the following convention.
Subset Naming Convention Templates
- SNOMED CT COVID-19 subsets: CV19-<category>-<subset>
- SNOMED CT COVID-19 subsets including subtypes of members: CV19-<category>-<subset>-withSubtypes
The table below shows some examples of subsets named using this convention.
| Examples of Named Subset | ||
|---|---|---|
| Category | Subset | Name |
| Provider and Facility Details (PFD) | Site of care subset | CV19-PFD-SiteOfCare |
Patient Demographics (PAT) | Marital status subset | CV19-PAT-MaritalStatus |
| Marital status subset including subtypes | CV19-PAT-MaritalStatus-withSubtypes | |
Clinical Assessment (ASS) | Symptoms subset | CV19-ASS-Symptoms |
| Symptoms subset including subtypes | CV19-ASS-Symptoms-withSubtypes | |
Subset Categories
The international SNOMED CT subsets have been organized into a number of categories, based on groupings of data elements that are likely to be recorded together. The five categories identified are:
Patient Demographics (PAT)
Clinical Assessment (ASS)
Tests and Investigations (INV)
Please follow the links above for information on the specific subsets within each category.
Subset Types
Some of the international subsets have been developed intensionally, while others have been developed extensionally. Please refer to the Practical Guide to Reference Sets, section 2.1.1. Subset Definitions for information on the difference between intensionally and extensionally defined subsets.
For each extensionally-defined subset, where the members have subtypes, we provide two versions:
- One subset which includes only the listed members. This version supports use cases (e.g. reporting, data integration) that require more abstract concepts.
- One subset which includes the members listed and all the subtypes of each of these members. This version supports use cases that may require more specific values (e.g. data collection where more clinical detail is required).
- Please note that this second type of subset is automatically generated from the corresponding subset with listed members. No manual curation has been performed, and therefore these subsets may contain concepts that are not relevant for the context of use.
Step 3 - Review and Adapt
The third step is to carefully review each international subset, and adapt it to meet your specific requirements. The subsets provided represent a collection of concepts that have been contributed from a range of SNOMED International Members. It is therefore important to review each subset member to:
- Remove any concepts that are not required for your use case
- Add any concepts missing from the subset that may be required for your specific use case
- Please note, if the concept you require is not included in the SNOMED CT Edition you are using, please follow the content request processes in each country. For more information, refer to the relevant SNOMED International Member page.
- SNOMED International National Release Centers and other authorized users may request additions or changes to the SNOMED CT International Edition via the SNOMED CT Content Request Service. For more information, refer to the CRS User Guide or contact info@snomed.org.
- Remove any concepts that are not included in the SNOMED CT versioned edition being used. This may involve
- Checking the effective time of each international concept to ensure that it is less than or equal to the International Version used by your local SNOMED CT edition
- Checking that any extension concepts are published in the SNOMED CT edition you are using
- Ensure that your subset aligns with best practice principles for subset creation.
- For example, this may involve checking that all members of the subset belong to a single hierarchy (in most cases), and that no two members subsumesubsume each other (in most cases).
- For more information, please watch our e-learning presentation on this topic - Subset Creation Principles.
Unpublished Content
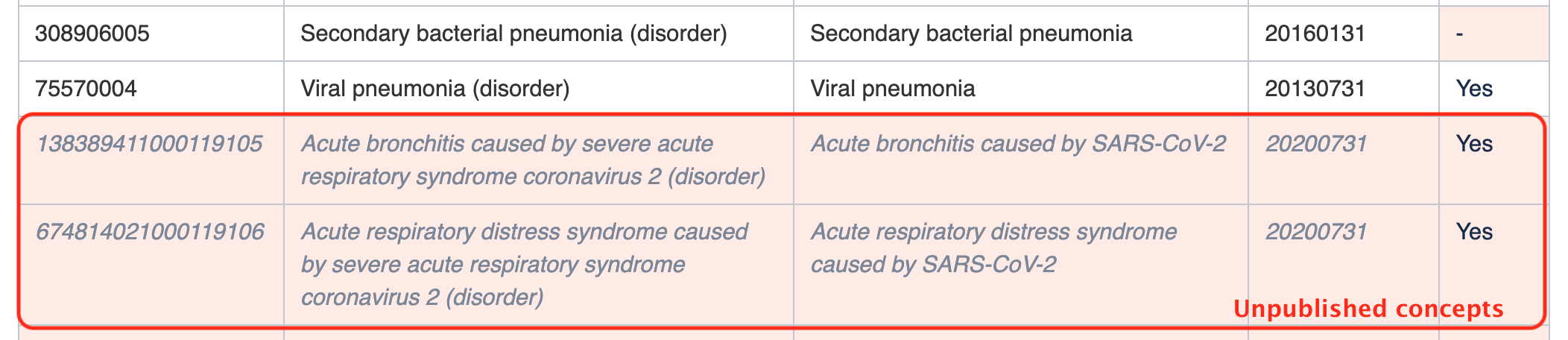
Please note that the subsets in this guide may include some unpublished content that is planned for a future SNOMED CT edition. This content should not be used until it has been officially published, as we cannot guarantee that the identifiers or terms will not change. For this reason, these unpublished concepts are not included in the downloadable versions of the subsets. Unpublished concepts are included in this guide to assist with planning (e.g. planning of national extensions).
Unpublished content is shown in this guide using grey, italic font on a pink background, as shown in the example below.
Extension Content
In response to an immediate need for COVID-19-specific concepts, various Member countries have developed concepts within their national SNOMED CT Extensions. Some of these extension concepts are documented in this guide, for the interest of other Members, see 4. Extension Content for COVID-19. Please note, however, that extension concepts should only be used when the module in which they are published is included in the implemented SNOMED CT Edition. SNOMED CT extension concepts are not included in the subsets downloadable from the Reference Set tool.
Feedback
SNOMED International welcomes comments on this guide and suggestions for new or updated content. Please use the Feedback button at the bottom of each page to send us your feedback.
Footnotes
Feedback