1 Introduction
1.1 Background
SNOMED CT terminology provides a common language that enables a consistent way of indexing, storing, retrieving, and aggregating clinical data across specialties and sites of care.
SNOMED International maintains the SNOMED CT technical design, the content architecture, the SNOMED CT content (includes the concepts table, the descriptions table, the relationships table, a history table, and ICD mappings), and related technical documentation.
1.2 Purpose
This document provides a summarized description of the content changes included in the July 2018 release of SNOMED Clinical Terms® (SCT) International Release.
It also includes technical notes detailing the known issues which have been identified. These are content or technical issues where the root cause is understood, and the fix has been discussed and agreed, but has yet to be implemented.
This document is available alongside the July 2018 International Edition release.
1.3 Scope
This document is written for the purpose described above and is not intended to provide details of the technical specifications for SNOMED CT or encompass every change made during the release.
1.4 Audience
The audience includes National Release Centers, WHO-FIC release centers, vendors of electronic health records, terminology developers and managers who wish to have an understanding of changes that have been incorporated into the July 2018 International Edition release.
2 Content Development Activity
2.1 Summary
Continuous quality improvement and enhancement of existing content is ongoing based on requests received via the Content Request System (CRS). A review of pre-coordination patterns has been undertaken to assist with the progression of CRS tickets that had been placed on hold. 35 patterns have had final decisions made and the corresponding CRS tickets progressed accordingly. Information on the pattern decisions may be found in the SNOMED CT Editorial Guide
The July 2018 International Release has seen a continuation of the work driven by contributions from: Kaiser Permanente Convergent Medical Terminology (CMT), Global Medical Device Nomenclature Agency (GMDNA) and Orphanet to add new content to SNOMED CT.
Additionally a process of Quality Improvement and project-driven changes have resulted in new content additions and enhancements to existing content detailed below.
Reminder Inactivation reason of LIMITED/WAS A will no longer be allowed for any content after the July 2018 release. The WAS A association refset will not be updated thereafter. Background In 2015, a proposal was made to inactivate 159083000 |WAS A (attribute)| relationship and stop updating the 900000000000528000|WAS A association reference set (foundation metadata concept)| at the Editorial Advisory Group. Since these recommendations were made, a formal proposal for the technical approach to batch updating the terminology was created and a notice of the proposed inactivation sent to the Community of Practice. The implementation of changes was postponed following feedback on utility for implementation and the potential impact to customers who were still using RF1. The matter was discussed again at the meeting of the Editorial Advisory Group in Bratislava in October 2017. Since the requirements and potential issues can be addressed by deriving such information from the RF2 release format, the recommendation is to proceed the decision after the July 2018 release. |
|---|
2.2 Content Quality Improvement
A total of 6494 new concepts were added
| SCT Statistics | New concept additions |
|---|---|
| SNOMED CT Concept (SNOMED RT+CTV3) | 6494 |
| Body structure (body structure) | 357 |
| Clinical finding (finding) | 1203 |
| Event (event) | 2 |
| Observable entity (observable entity) | 60 |
| Organism (organism) | 182 |
| Pharmaceutical / biologic product (product) | 3053 |
| Physical object (physical object) | 88 |
| Procedure (procedure) | 659 |
| Qualifier value (qualifier value) | 224 |
| Record artifact (record artifact) | 5 |
| SNOMED CT Model Component (metadata) | 10 |
| Situation with explicit context (situation) | 64 |
| Social context (social concept) | 27 |
| Specimen (specimen) | 2 |
| Staging and scales (staging scale) | 21 |
| Substance (substance) | 537 |
A total of 32889 changes were made to existing content
| SCT Improvement Statistics to Existing Concepts | Number |
|---|---|
| Total number of changes | 32889 |
| Change to stated concept definition | 20126 |
| Change to inferred concept definition | 27970 |
| Change in concept definition status from Primitive to Sufficiently Defined | 7997 |
| Description change | 10691 |
| Inactivated concepts | 6955 |
| Inactivated descriptions | 9256 |
| Reactivated concepts | 16 |
2.2.1 Anatomy
Advance Notice Planned inactivation of 123005000 |Part of (attribute)| attribute and relationships in the January 2019 release |Part of| relationships have only been included in the inferred relationship file. They are not actively maintained because they are additional relationships rather than defining relationships. The new anatomy concept model has introduced different kinds of 'part of' relationships, e.g. |All or part of|, |Constitutional part of|, |Lateral half of|, |Regional part of|, and |Systemic part of|. The modeling Advisory Group has recommended inactivation of 123005000 |Part of (attribute)| attribute and relationships in the January 2019 release. It will be replaced by a new attribute |Proper part of| to represent defining relationships where 'part is not equal to whole'. The new anatomy model will be implemented in future releases when the required description logic enhancements are implemented. |
|---|
New anatomy concepts: 357
- 333 new body structure concepts.
- 24 new morphologic abnormality concepts.
- 276 new lateralized body structure concepts.
- There are now over 5,600 lateralized concepts in SNOMED CT and over 300 concepts are new lateralized findings, disorders and procedures.
Updates have been made to 109 concepts to conform with the new editorial guidance on the naming convention for digits of hand and foot.
- Added 196 new descriptions.
- Inactivated 174 descriptions.
Updates have been made to concepts for skin and/or subcutaneous tissue to conform to the editorial guidance on the naming convention for conjunction and disjunction.
- Added 84 new descriptions.
- Inactivated 30 descriptions.
2.2.2 Clinical finding
New concepts for clinical findings and disorders: 1203
Work completed on the following Content Projects:
IHTSDO-451 - Has definitional manifestation.
- IHTSDO-592 and IHTSDO-381 - Inactivate 91138005|Mental retardation (disorder)| and replace with 110359009|Intellectual disability (disorder)| with change to 281 descendant concepts.
- IHTSDO-955 - Remodel Talipes Valgus.
Work commenced on the following Content Projects:
- IHTSDO-703 - Inactivation of 'unilateral' concepts. Work on the inactivation of unilateral concepts and the creation of left and right replacement concepts continued this release. The focus areas were: Cleft lip, Femoral hernia, Inguinal hernia, Kidney disorder/procedure, Orchidectomy, Orchipexy, Lymphangiography, Vascular imaging procedures. In the areas of cleft lip and hernia additional modelling improvements were also made. Further work is still required to complete the inactivation of unilateral concepts.
- IHTSDO-614 - Rework hierarchy 441457006|Cyst (disorder)| work on this project continues.
- IHTSDO- 393 Diabetes Complications - Work has begun on updating and modeling content relating to the diagnosis and management of patients with diabetes mellitus. Initial work relates to diabetic complications where the editorial guidance on combined disorders and complications is being applied. A small task and finish group consisting of representatives of the multidisciplinary care team will be reviewing existing content and advising on new content to ensure that SNOMED CT is fit for the purposes of supporting the electronic health care record for this group of patients.
- IHTSDO-114 - 183 out of 585 descendants of 128294001 |Chronic inflammatory disorder (disorder)| have been remodeled with stated relationship changed from Associated Morphology = Chronic inflammation (morphologic abnormality) to Associated morphology (attribute) = Chronic inflammatory morphology (morphologic abnormality), made Fully Defined, modeled to primitive parent, addition of Clinical course (attribute) = Chronic (qualifier value) and other relationships added or grouped as necessary. Stated children of inferred subtypes of 128294001 |Chronic inflammatory disorder (disorder)| have started to be addressed following the pattern above, and the remainder are now part of the QI work for inclusion in a future release. 249 inferred concepts require a stated relationship change from Associated Morphology = Chronic inflammation (morphologic abnormality) to Associated morphology (attribute) = Chronic inflammatory morphology (morphologic abnormality).
Other content quality improvements:
- For the January 2018 release the synonym of "occlusion" was inactivated from 26036001|Obstruction (morphologic abnormality) and added to 50173008|Complete obstruction (morphologic abnormality). 415582006|Stenosis (morphologic abnormality)| became a descendant of 25659002|Narrowed structure (morphologic abnormality). These changes resulted in a further 133 released concepts being reviewed and/or changed for the July 2018 release.
Added a new sufficiently defined concept of 763597000|Hereditary ataxia (disorder)| resulting in changes to 40 concepts.
- Hierarchy review/remodel descendants of 248595008|Sputum (finding)| with changes to 59 concepts.
- Review/remodel "Progressive" disorders with changes to approximately 200 concepts. A new clinical course qualifier value has been created: 255314001|Progressive (qualifier value)| as a subtype of 90734009|Chronic (qualifier value)|. Can be used to fully define progressive disorders using 246454002|Occurrence (attribute)| = progressive (qualifier value)
- Review/remodel of "Acquired" disorders with changes to 697 concepts. A new period of life has been created: 767023003|Period of life beginning after birth and ending before death (qualifier value)|. This allows for a distinction to be made between acquired vs. congenital concepts and also permits existing ‘acquired’ concepts to be sufficiently defined.
- Addition of Bipolar disorder concepts which include the most recent episode information.
36 new concepts added for genetic diseases which were not part of the Orphanet project.
Remodeling and re-terming of 110288007 |Joint tenderness (finding)|. Redefined Joint tenderness as IS_ A Tenderness (finding) + finding site=joint structure and all of the specific joint tenderness concepts using the same pattern. Renamed all joint tender(ness) concepts as Tenderness of x joint (finding).
Inactivated 202936005 |Ganglion and cyst of synovium, tendon and bursa (disorder) and used an analogous approach to inactivate the descendants with new replacement concepts.
Remodel 63491006 |Intermittent claudication (finding)| hierarchy. Added due to 86341008 |Vascular insufficiency (disorder)|. Removed parent of 22253000 |Pain (finding)| and added parents of 427935006 |Pain relief by rest (finding)| and 427341007 |Pain provoked by exertion (finding)|.
Review and remodel 11 viral disorder and related concepts.
- 45 new clinical finding/disorder concepts added for Dentistry.
Inactivated out of scope |Allergy to X (disorder)| concepts, as identified by the Allergy/Hypersensitivity and Intolerance Clinical Reference Group.
Review and remodel of 520 fracture of lower limb concepts using a proximal primitive parent and creating stated defining relationships for those which were previously inferred relationships.
Intussusception disorder concepts were remodeled using the proximal primitive parent modeling approach with two relationship groups capturing the invagination and obstruction morphologic abnormalities of this disorder. The concept Intussusception (morphologic abnormality) was inactivated as a duplicate of Invagination (morphologic abnormality).
2.2.3 Convergent Medical Terminologies (CMT)
New CMT concepts: 215
These were added across 3 domains:
- CMT Cardiology
- CMT Hematology oncology
- CMT Musculoskeletal
2.2.4 Procedure Hierarchy
New concepts for procedure hierarchy: 659
Diagnostic imaging - 32 requests were received for new imaging procedure concepts with various imaging modalities.
Work completed on the following Content Projects:
- IHTSDO-688 - Improvement in the modelling of the Brachytherapy sub-hierarchy.
- IHTSDO-688 - Improvement in the modelling of the Brachytherapy sub-hierarchy.
Other content quality improvements:
Implemented changes related to 208 client requests mainly in the area of laboratory procedure and microbiology reporting. Changes are implemented in the following hierarchies: Organism (including creation of 133 new organism concepts), Specimen, Evaluation procedure, Finding, Substance, and Technique.
Updated naming for Streptococcus pneumoniae serotypes by specifying the nomenclature system (Danish vs. American) which resulted in changes for 214 concepts (including addition, inactivation or change in description) in the following hierarchies: Organism, Substance, and Evaluation procedure.
2.2.5 Collaboration/Harmonization Projects
2.2.5.1 Orphanet
As a result of collaboration with Orphanet (http://www.orpha.net/consor/cgi-bin/index.php) 473 new SNOMED CT concepts have been created to represent rare diseases in the disorder hierarchy.
2.2.5.2 Global Medical Device Nomenclature Agency (GMDNA)
64 new SNOMED CT concepts created and mapped in the Physical object hierarchy to support the GMDN collaboration agreement.
2.2.6 Event
New concepts added: 2
Work commenced on the following Content Projects:
- IHTSDO-1071 - Vehicle accident (event) - Inactivation of over 200 transport accidents from Event hierarchy.
2.2.7 Qualifier Value
New concepts added: 224
Content quality improvements:
- Several new concepts have been added for Pathological process (qualifier value) to support the upcoming revisions to the allergy model.
Replaced existing ambiguous top-level concept 258666001 |Unit (qualifier value)| with new 767524001 |Unit of measure (qualifier value)| concept. To support modeling of clinical drug concepts in the 763158003 |Medicinal product (product)| hierarchy, created new 767525000 |Unit (qualifier value)| concept as descendant of 259026009 |Miscellaneous unit (qualifier value)|.
- 308912000|Agencies and organizations (qualifier value)| and subtypes moved to a National extension, 66 concepts inactivated from the International release.
- Several new concepts have been added for Pathological process (qualifier value) to support the upcoming revisions to the allergy model.
2.2.8 Organism
New organism concepts added: 182
Work commenced on the following Content Projects:
- IHTSDO-455 - Organism Life Cycles - The project was undertaken to clarify the meaning of organism concepts that are likely values for laboratory reports related to parasitic and certain other infections. Concept descriptions and definitions were edited for concepts such as 609326000 | Strongyloides stercoralis larval form (organism) |, previously placed as subtypes of both 421727006 | Phylum Nemata (organism) and 278306005 | Life-cycle form (organism) |, so that their status as organisms was made clear. This project altered 320 concepts representing various taxonomic groups including phylum Nemata, class Cestoda, class Trematoda, protozoa, Fungi, yeasts and viruses. A limited number of concepts that could not be related to a specific organism (e.g. 278306005 | Life-cycle form |) were inactivated. In cases where a diagnosis is made by identifying an organism structure (e.g. 284688000 | Fungal hyphae |), the organism concept was inactivated and referred to a finding concept “404507002 | Hyphae of Kingdom Fungi detected (finding).
Other content quality improvements:
- Updated naming for Streptococcus pneumoniae serotypes by specifying the nomenclature system (Danish vs. American) which resulted in changes for 214 concepts (including addition, inactivation or change in description) in the following hierarchies: Organism, Substance, and Evaluation procedure.
2.2.9 Pharmaceutical / biologic product
For further details on the planned changes in this area, please refer to the Drug Model Working Group Directory section. Please note, you may have to register for Confluence user account in order to access this project and the relevant links below. |
The following changes will be included in the 2018-July International Release.
| New hierarchies |
|
|---|---|
| New semantic tags |
|
New attribute types |
|
| Grouper concepts representing disposition, intended site, and/or structure |
New high level grouper concepts created as an incremental improvement with additional subtypes to be added in future releases:
|
| Product role concepts |
|
Medicinal product (MP) concepts |
|
| Medicinal product form (MPF) concepts |
|
Clinical drug (CD) concepts | CD-containing precisely concepts sufficiently defined by modeling to proximal primitive concept, adding defining attributes, and updating descriptions per editorial guidelines (n≈ 4850). Includes the following attributes:
|
| Documentation |
|
2.2.10 Veterinary Extension
15 concepts were moved to the Veterinary Extension.
2.2.11 Situation with Explicit Context
New concepts added : 64
143 concepts using the pattern 'OFFERED' or 'NOT OFFERED' were inactivated.
Addition of new concepts for transplant donor and recipient Epstein Barr virus status.
Work continuing on the following Content Projects:
- IHTSDO-703 - Inactivation of 'unilateral' concepts. Work on the inactivation of unilateral concepts and the creation of left and right replacement concepts continued this release.
- IHTSDO-703 - Inactivation of 'unilateral' concepts. Work on the inactivation of unilateral concepts and the creation of left and right replacement concepts continued this release.
2.2.12 Social Context
26 new concepts were added as a subtype of 14679004|Occupation (occupation)|.
2.2.13 Assessment Scale
Review of 11 of assessment scale concepts for duplicates or correctness.
Addition of 96 assessment scale, observable entity and procedure concepts to support use of assessment scales.
2.2.14 Substances
The following changes will be included in the 2018-July International Release.
| Disposition groupers |
|
|---|---|
| Structural groupers |
|
| Chemical element groupers |
|
| Combined groupers |
|
| Is modification of attribute |
|
| Role groupers |
|
| General content updates |
|
Documentation |
|
Other content quality improvements:
Inactivation of 417881006|Radiopharmaceutical agent (substance)| which was a primitive concept with three stated subtypes and not a substance but a role of a substance. After the inactivation, there was a hierarchy review starting at 33638001|Isotope (substance)| involving approximately 850 concepts.
Creation of new terming document, use of INN terming where possible. Alignment of content with new terming
Removal of caret "^" for superscript
For example:
Before = 24511001|Technetium Tc^99m^ succimer (substance)
After = 24511001|Technetium (99m-Tc) succimer (substance)
Substances were moved under more specific supertypes where possible. Removal of formatting carets
Identified nuclear medicine procedures using a "substance grouper" as an attribute value but refer to a specific substance in the FSN. Remodeled with a specific substance approximately 93 concepts
Concept 371572003|Nuclear medicine procedure (procedure)| has been sufficiently defined. A hierarchy review has been started to review terming, use of correct radioisotopes, and inactivation of concepts containing additional context outside of the actual procedure.
For further details on the planned changes in this area, please refer to the Substances project. Please note, you may have to register for Confluence user account in order to access this project and the relevant links above. |
2.3. Internal Quality Improvement
2.3.1 Logic Profile Enhancements
The first release of the OWL reference sets
As an essential part of SNOMED CT Logic Profile Enhancements, two OWL refsets have been developed to represent logic definitions following the international standard of OWL 2 Web Ontology Language.
The OWL Ontology reference set includes essential metadata information about an ontology, such as, namespaces, ontology URI and ontology version URI.
The OWL Axiom reference set includes only property axioms, such as property chains, transitive properties, and reflexive properties, that cannot be fully represented by the RF2 stated and inferred relationship files. The limited scope of the first release is a response to the Community of Practice Consultation happened early this year, which allows sufficient time for users to develop and update their tooling and systems.
The content from the OWL reference sets must be included for all applications that utilise the description logic (DL) reasoning services to ensure correct inferences. These property axioms are required for classification by DL reasoners to support the drug, substance, and anatomy projects and other clinical content depend on them. Extensions can update the refsets by following the OWL reference sets specification.
Following the recommendation from the Modeling Advisory Group, the full representation of all logical definitions and other key DL features, such as additional axioms and GCIs (General Concept Inclusion), will be included in the OWL axiom refset in the January 2019 release. A reference implementation by SNOMED International was demonstrated at the April business meeting. All software systems implemented DL reasoning services need to be updated to use the OWL axiom refset that is the only source of truth for logic definitions from 2019.
Updates to the RF2 relationship files
There are no changes to the stated relationship file for the July 2018 release. However, the stated relationship file must be used in conjunction with the OWL refsets for the DL reasoning services.
In the January 2019 release, the stated relationship file will be replaced by the OWL axiom refset that includes all logical definitions and other features specified in the SNOMED CT logic profile specification. The stated relationship file will NOT be included in the international release; however, it may still be available on request to support migration to the OWL axiom refset.
The inferred relationship file will maintain the same format and structure, though it is no longer containing all necessary and sufficient conditions. The inferred relationship file is represented in Necessary Normal Form for distribution of relationships. It is a collection of all the necessary conditions and represents a subset of the full semantics from the 2018 July release and onwards. Most users will benefit from the improvements in the inferred relationships without requiring changes to their existing systems.
A set of documentations has been developed to support the Logic Profile Enhancements.
- SNOMED DL Profile Enhancements - https://docs.google.com/document/d/1tqNEA6S4fEF4fgj15OPabYA2E0VTz8epxvRRwczKizQ/edit?usp=sharing
- SNOMED CT Logic Profile Specification - http://snomed.org/lps
- SNOMED CT OWL Guide (OWL Refsets specification) - http://snomed.org/owl
- Necessary Normal Form for Inferred Relationships - https://docs.google.com/document/d/1dt0r0aetwpwmHOfiT9wt0EVukVLRvVjXYUn_vq-QhIM/edit?usp=sharing
- Snomed OWL Toolkit - https://github.com/IHTSDO/snomed-owl-toolkit
- Classifying SNOMED CT using the Snomed OWL Toolkit - https://youtu.be/-91egY9mJqA
- Creating an OWL file containing SNOMED CT - https://youtu.be/sfFbMMioA_4
2.3.2 Machine Readable Concept Model (MRCM)
14 new MRCM rules and 10 updates to the existing MRCM in the MRCM refsets and authoring platform.
- New MRCM for new attribute 738774007 |Is modification of|
- New MRCM for new attribute 762951001 |Has ingredient|
- New MRCM for new attribute 762949000 |Has precise active ingredient|
- New MRCM for new attribute 411116001 |Has manufactured dose form|
- New MRCM for new attribute 763032000 |Has unit of presentation|
- New MRCM for new attribute 733928003 |All or part of|
- New MRCM for new attribute 733931002 |Constitutional part of|
- New MRCM for new attribute 733933004 |Lateral half of|
- New MRCM for new attribute 733930001 |Regional part of|
- New MRCM for new attribute 733932009 |Systemic part of|
- New MRCM for new attribute 766952006 |Count of base of active ingredient|
- New MRCM for new attribute 766954007 |Count of base and modification pair|
- New MRCM for new attribute 766953001 |Count of active ingredient|
- New MRCM for new attribute 766939001 |Plays role|
- Change the range for attribute 246514001 |Units| from |Unit| to |Unit of measure|
- Change the range for attribute 733722007 |Has concentration strength denominator unit| from |Unit| to |Unit of measure|
- Change the range for attribute 733725009 |Has concentration strength numerator unit| from |Unit| to |Unit of measure|
- Change the range for attribute 732947008 |Has presentation strength denominator unit| from |Unit| to |Unit of measure|
- Change the range for attribute 732945000 |Has presentation strength numerator unit| from |Unit| to |Unit of measure|
- Expand the range of 726542003 |Has disposition| to include 726711005 |Disposition|
- Expand the range of 704319004 |Inheres in| to include << 125676002 |Person (person)|
- Expand the range of 118170007 |Specimen source identity| to include << 260787004|Physical object|
- Expand the range of 704321009|Characterizes| to include << 71388002 |Procedure|
2.4 SNOMED CT derived products
2.4.1 ICD-10 map
The SNOMED CT to the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (© World Health organisation 1994) 2016 Version map (SNOMED CT to ICD-10 Map) is included in the SNOMED CT International release as a Baseline. The SNOMED CT to ICD-10 Map was created to support the epidemiological, statistical and administrative reporting needs of SNOMED International member countries and WHO Collaborating Centres.
The SNOMED CT to ICD-10 Map is released in Release Format 2 (RF2) only. It is located in the file der2_iisssccRefset_ExtendedMapFull_INT_20180731.txt, which is in the Map folder under Refset, in each of the three RF2 Release Type folders.
The SNOMED CT to ICD-10 Map is released as Refset 447562003 |ICD-10 complex map reference set (foundation metadata concept)|.
2.4.1.1 Content development activity summary
The map is a directed set of relationships from SNOMED CT source concepts to ICD-10 target classification codes. The SNOMED CT source domains for the MAP are limited to subtypes of 404684003 |clinical finding|, 272379006 |event| and 243796009 |situation with explicit context|. The target classification codes are ICD-10 2016 release.
2.4.1.2 Mapped content for July 2018
The map provided for the July 2018 release has been updated, and now represents a complete map from SNOMED CT International release to ICD-10 2016 version.
- 1278 new concepts added
We would welcome feedback on any issues that users of the map may detect when using the map. Issues should be submitted via mapping@snomed.org
2.4.1.3 Technical Guide Exemplars
The Technical Guide Exemplars document has now been moved from the International Edition release package to a Confluence page. This page can be found as part of the ICD-10 Mapping Technical Guide (see Appendix B), which is hosted here: http://snomed.org/icd10map
2.4.2 ICD-O Map
There are 5 updates for the ICD-O Morphological abnormality map in July 2018 release.
2.4.3 SNOMED CT to OWL conversion and classification
The repository containing the toolkit enabling simple SNOMED CT to OWL conversion and classification can be found here, including documentation on its use:
https://github.com/IHTSDO/snomed-owl-toolkit
Please contact SNOMED International at support@snomed.org if you would like to provide any feedback on ways to extend and improve the new toolkit.
3 Technical notes
3.1 Known Issues
Known Issues are content or technical issues where the root cause is understood, and the resolution has been discussed and agreed but has yet to be implemented. This can be due to a number of reasons, from lack of capacity within the current editing cycle, to the risk of impact to the stability of SNOMED CT if the fix were to be deployed at that stage in the Product lifecycle.
For the Snomed CT July 2018 International edition, the following Known Issues were identified, and agreed to be resolved in the next editing cycle (to be published in July 2018):
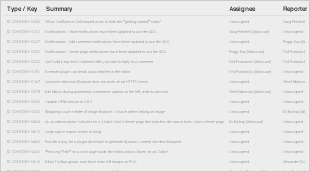
 3.2 Resolved Issues
3.2 Resolved Issues
Resolved issues are Known Issues which were not fixed as part of the previous release lifecycle, but which have now been resolved in the latest release - in this case the July 2018 International Edition. They can also be issues found during the Alpha and Beta testing of the current release, which were resolved before the final deployment of the associated Member release. Finally they can be issues which were reported or found during the testing phase, but which have been closed without any action taken.
The Resolved Issues for the Snomed CT July 2018 International edition can be found here:

3.3 Technical updates
3.3.1 RF2 package format
For future reference, the RF2 package convention dictates that it contains all relevant files, regardless of whether or not there is content to be included in each particular release. Therefore, the package contains a mixture of files which contain both header rows and content data, and also files that are intentionally left blank (including only a header record). The reason that these files are not removed from the package is to draw a clear distinction between
- ...files that have been deprecated (and therefore removed from the package completely), due to the content no longer being relevant to RF2 in this or future releases, and
- ...files that just happen to contain no data in this particular release (and are therefore included in the package but left blank, with only a header record), but are still relevant to RF2, and could therefore potentially contain data in future releases.
This allows users to easily distinguish between files that have purposefully been removed or not, as otherwise if files in option 2 above were left out of the package it could be interpreted as an error, rather than an intentional lack of content in that release.
3.3.2 Introduction of two new refsets - OWLAxiom and OWLOntology
Please see section "2.3.1 Logic Profile Enhancements" above for full details of the reason behind the introduction of these new refsets. The technical implication is that two new files will be included in each section of the International Release package (Full, Snapshot and Delta):
- sct2_sRefset_OWLOntologyFull_INT_20180731.txt
- sct2_sRefset_OWLAxiomFull_INT_20180731.txt
- sct2_sRefset_OWLOntologySnapshot_INT_20180731.txt
- sct2_sRefset_OWLAxiomSnapshot_INT_20180731.txt
- sct2_sRefset_OWLOntologyDelta_INT_20180731.txt
- sct2_sRefset_OWLAxiomDelta_INT_20180731.txt
The naming convention was decided upon in order to best describe the content of each refset file. It was thought to be misleading to include ’stated’ at the start of the file names because the OWL Ontology Refset contains only metadata for an ontology, which is always the same no matter whether the ontology contains axioms before or after classification. In addition, the word 'stated' is redundant to 'axiom'. Finally, the 'sct2' filetype is utilised to indicate that the axioms are not a derivative product.
The decision was taken to place the files in the "Terminology" folder in the release package (as opposed to the "Refset" folder), because these files are designed to eventually replace the Stated relationship files, and therefore contain core content which needs to be included in the Terminology folder.
3.3.3 Replacement of OWL conversion script with a link to the open source directory
In January 2017 the original OWL conversion script (a.k.a. the "Spackman OWL script") was removed from the International Edition package, and has since been published as a separate artefact alongside each Release.
Because this script does not recognize the two new OWL refsets (see section 3.3.2 of these Release Notes), the Terminology Release Advisory Group has determined that the script should no longer be distributed.
Going forward, Release Notes will now include a link to the new open source OWL conversion toolkit that can be found in the following repository (including documentation on its use):
https://github.com/IHTSDO/snomed-owl-toolkit
Please contact SNOMED International at support@snomed.org if you would like to provide any feedback on ways to extend and improve the new toolkit.
3.3.4 Deprecation of the Technical Guide Exemplars document from the International Edition release package
The Terminology Release Advisory Group has confirmed that there should be no impact from removing this almost entirely static document from the International Edition release package, and hosting it instead in a Confluence Page. From the July 2018 International release then, we will deprecate this file (doc_Icd10MapTechnicalGuideExemplars_[date].xlsx) from the Release package, and instead provide a link in the Release Notes to the new Confluence Page. Please see section 2.4.1.3 above for the relevant link.
3.3.5 Early visibility of impending change in the January 2019 International edition
Please see the following page for details of all upcoming changes planned for January 2019 and beyond: Early Visibility - Planned changes to upcoming SNOMED International Release packages
 3.3.6 IMPORTANT CHANGE in the January 2019 International Edition
3.3.6 IMPORTANT CHANGE in the January 2019 International Edition
Replacement of the Stated Relationship files with the new OWL Axiom refset files
In the January 2019 release, the stated relationship file will be replaced by the OWL Axiom refset files, that will include all logical definitions and other features specified in the SNOMED CT logic profile specification. The stated relationship file will NOT be included in the international release; however, it may still be available on request to support migration to the OWL Axiom refset.
| Users should carefully analyse any potential impact to their systems (upload routines, etc) and make provisions for these changes urgently (if not already done), in order to prevent any issues when these changes come into effect in January 2019. Please contact SNOMED International at support@snomed.org with “OWL Axiom refset files implementation question” in the subject line. |
The inferred relationship file will maintain the same format and structure, though it will no longer contain all necessary and sufficient conditions. The inferred relationship file is represented in Necessary Normal Form for distribution of relationships. It is a collection of all the necessary conditions and represents a subset of the full semantics from the 2018 July release and onwards. Most users will benefit from the improvements in the inferred relationships without requiring changes to their existing systems.
A set of documentations has been developed to support the Logic Profile Enhancements.
- SNOMED DL Profile Enhancements - https://docs.google.com/document/d/1tqNEA6S4fEF4fgj15OPabYA2E0VTz8epxvRRwczKizQ/edit?usp=sharing
- SNOMED CT Logic Profile Specification - http://snomed.org/lps
- SNOMED CT OWL Guide (OWL Refsets specification) - http://snomed.org/owl
- Necessary Normal Form for Inferred Relationships - https://docs.google.com/document/d/1dt0r0aetwpwmHOfiT9wt0EVukVLRvVjXYUn_vq-QhIM/edit?usp=sharing
- Snomed OWL Toolkit - https://github.com/IHTSDO/snomed-owl-toolkit
- Classifying SNOMED CT using the Snomed OWL Toolkit - https://youtu.be/-91egY9mJqA
- Creating an OWL file containing SNOMED CT - https://youtu.be/sfFbMMioA_4
3.3.7 Document links
All links provide information that is correct and current at the time of this Release. Updated versions may be available at a later date, but if so these will need to be requested from the relevant SNOMED International teams.
NOTE: To access any of the links in the pdf document, please visit the Release Notes online here: |
Approvals
|
Draft Amendment History
|
