1 Introduction
1.1 Background
SNOMED CT terminology provides a common language that enables a consistent way of indexing, storing, retrieving, and aggregating clinical data across specialties and sites of care.
SNOMED International maintains the SNOMED CT technical design, the content architecture, the SNOMED CT content (includes the concepts table, the descriptions table, the relationships table, a history table, and ICD mappings), and related technical documentation.
1.2 Purpose
This document provides a summarized description of the content changes included in the July 2017 release of SNOMED Clinical Terms® (SCT) International Release.
It also includes technical notes detailing the known issues which have been identified. These are content or technical issues where the root cause is understood, and the fix has been discussed and agreed, but has yet to be implemented.
This document is available alongside the July 2017 International Edition release.
1.3 Scope
This document is written for the purpose described above and is not intended to provide details of the technical specifications for SNOMED CT or encompass every change made during the release.
1.4 Audience
The audience includes National Release Centers, WHO-FIC release centers, vendors of electronic health records, terminology developers and managers who wish to have an understanding of changes that have been incorporated into the July 2017 International Edition release.
2 Content Development Activity
2.1 Summary
Continuous quality improvement and enhancement of existing content is ongoing based on requests received via the Content Request System (CRS).
As well, the July 2017 International Release has seen a continuation of the work driven by contributions from: Kaiser Permanente Convergent Medical Terminology (CMT), Global Medical Device Nomenclature Agency (GMDNA), World Health Organization (WHO) ICD-11 Beta, International Classification for Nursing Practice ( ICNP®) and Orphanet to add new content to SNOMED CT.
Additionally, member-identified priority projects, have driven enhancements in the anatomy hierarchy, while other project-driven changes have resulted in new content additions and enhancements to existing content detailed below.
 2.2 Content Quality Improvement
2.2 Content Quality Improvement
Summary of changes
In total: 10770 new concepts were authored with an additional 15993 changes made to existing concepts.
| SCT Statistics | New concept additions |
|---|---|
| SNOMED CT Concept (SNOMED RT+CTV3) | 10773 |
| Body structure (body structure) | 5994 |
| Clinical finding (finding) | 2144 |
| Event (event) | 10 |
| Observable entity (observable entity) | 25 |
| Organism (organism) | 123 |
| Pharmaceutical / biologic product (product) | 83 |
| Physical object (physical object) | 224 |
| Procedure (procedure) | 665 |
| Qualifier value (qualifier value) | 735 |
| Record artifact (record artifact) | 13 |
| SNOMED CT Model Component (metadata) | 76 |
| Situation with explicit context (situation) | 198 |
| Social context (social concept) | 2 |
| Specimen (specimen) | 27 |
| Staging and scales (staging scale) | 19 |
| Substance (substance) | 435 |
2.2.1 Anatomy
- Two new association reference sets have been developed and included in this release. The new refsets will help to identify missing concepts, ensure quality of concept modeling and enable systematic application of naming convention for S, E, and P concepts. refsets represent the associations between anatomy Structure, Entire and Part:
- Anatomy Structure and Entire Association Reference Set
- Anatomy Structure and Part Association Reference Set
- The Lateralizable Body Structure reference set has been updated in the SNOMED International refset tool as follows:
- 3728 new refset members added and 5 refset members inactivated for the July 2017 release
- 3728 new refset members added and 5 refset members inactivated for the July 2017 release
- New content has been added as required for the development of a new concept model for anatomy, as well as alignment with other anatomy terminology standards as follows:
- 5,768 new concepts; 5,427 are Entire concepts which have been added to ensure the completeness of SEP model.
- 200 (approx.) new lateralized body structures and
- 800 new lateralized procedures and disorders
- 11 new enthesis concepts added to support the revision of sub-hierarchy of 23680005|Enthesopathy (disorder)| which were incorrectly modeled by two body structures
2.2.2 Clinical finding
- Added 13 new concepts for clinical findings and morphological abnormalities used to represent laboratory findings
- Crohn's disease - review and/or remodelling of 55 concepts plus addition of 2 new concepts
2.2.3 Convergent Medical Terminologies (CMT)
A total of 668 new concepts were added as follows:
- CMT Cardiology - 12 concepts
- CMT Emergency Department - 12 concepts
- CMT Endocrine - 6 concepts
- CMT Hematology Oncology - 6 concepts
- CMT Hx and Family Hx - 82 concepts
- CMT Injuries - 14 concepts
- CMT Mental Health - 31 concepts
- CMT Miscellaneous - 104 concepts
- CMT Musculoskeletal - 272 concepts
- CMT Neurology - 2 concepts
- CMT Obstetrics and Gynecology - 2 concepts
- CMT Ophthalmology - 52 concepts
- CMT Orthopedics Extremity Fractures - 2 concepts
- CMT Pediatrics - 2 concepts
- CMT Radiology - 66 concepts
- CMT Top 2500 - 3 concepts
2.2.4 Diagnostic Imaging Procedures
- 342 (approx.) including:
- MRI 120
- CT 102
- X-ray 12
- Venography 2
- Fluoroscopy 31
- Ultrasonography guided 59
- Ultrasonography 16
- Laterality concepts across MRI, CT, X-ray and ultrasonography and interventional radiology
2.2.5 Disorders
2.2.5.1 Orphanet
- As a result of collaboration with Orphanet (http://www.orpha.net/consor/cgi-bin/index.php) 485 new concepts have been created to represent rare diseases in the disorder hierarchy.
2.2.5.2 ICD-11 Harmonization plan
- 1200 ICD-11 concepts added from the Mortality and Morbidity Linearization
2.2.5.3 Revision of 12962009|Histoplasmosis (disorder) - re-work hierarchy to fix inheritance issues and ensure consistent use of attributes.
2.2.6 LOINC
- 114 new concepts were added to support the mapping of LOINC Parts to SNOMED CT concepts for the LOINC - SNOMED CT Cooperation Project. This included properties, techniques, population of cells, cells, etc.
2.2.7 Morphologic Abnormality
114 new morphologic abnormality concepts added and then mapped to International Classification of Diseases for Oncology Third Edition (ICD-O First Revision, or ICD-O-3.1) 2011
2.2.8 Observables
- Renamed 5 “X detail observables (observable entity)” concepts to “X observable” for clarity and retired 1 “X detail observable” as ambiguous. Retired 1 ambiguous Observable entity grouper concept and 1 related ambiguous Clinical finding concept.
2.2.9 Organisms
- 605 changes were made to terms which used an incorrect case in a Salmonella serovar name.
- 170 changes were made to support Microbiology reporting. The majority of these were new organism concepts.
Of these, 532 term updates were made in the Organism hierarchy and 73 term replacements were made in other hierarchies (Procedure, Substance, Product, Disorder).
2.2.10 Pharmaceutical / biologic product
Added >15 new descriptions with "medication" where "drug" was already used
For further details on the planned changes in this area, please refer to the Drug Model Working Group Directory section. Please note, you may have to register for Confluence user account in order to access this project. |
Summary of changes
| New hierarchies |
|
|---|---|
| New semantic tags |
|
New attribute types |
|
| Attribute description changes |
|
| Grouper |
|
| Medicinal product (MP) |
|
| Medicinal product form (MPF) |
|
| Clinical drug (CD) |
|
2.2.11 Physical objects
- 218 new device concepts added.
2.2.12 Procedures
- Revision of hierarchy 23968004|Colectomy (procedure) and subtypes
- Laterality requests for diagnostic imaging across MRI, CT, X-ray and ultrasonography and interventional radiology
2.2.13 Social concept
No significant changes for this release.
2.2.14 Specimen
- 30 new concepts were added
2.2.15 Substances
- Summary of changes:
- New hierarchy added with semantic tag: Disposition (disposition) 726711005
- New attribute added: Has disposition(attribute) 726542003
- Concepts containing disjunctives were inactivated, e.g:
- Antigen of Neisseria meningitidis A + Y (substance)
- Butyrylcarnitine + isobutyrylcarnitine (substance)
- Immunoglobulin A antibody to Bordetella pertussis filamentous hemagglutinin + Bordetella pertussis toxin (substance)
- Concepts containing brand names were Inactivated e.g:
- Horlicks drink (substance)
- Ovaltine drink (substance)
For further details on the planned changes in this area, please refer to the Substances project. Please note, you may have to register for Confluence user account in order to access this project. |
2.2.16 Foundation metadata component
Descriptions that are inactivated in the International Edition will use one of the following four values:
- | Not semantically equivalent component (foundation metadata concept) | - new concept
- | Outdated component (foundation metadata concept) |
- | Erroneous component (foundation metadata concept) |
- | Non conformance to editorial policy component (foundation metadata concept) | - new concept.
 2.3. Internal Quality Improvement
2.3. Internal Quality Improvement
2.3.1 Case significance indicator
In order to align in accordance with the RF2 specification, a technical batch change was made to the case significance value for over 760,000 descriptions.
- Changed from 900000000000020002|Only initial character case insensitive (core metadata concept)| to 900000000000448009|Entire term case insensitive (core metadata concept)|.
For further details please refer to the document linked here: IHTSDO-950 Update of case significance assignment following the RF2 specification
2.3.2 OWL axiom reference set
Following the recommendation from Modeling Advisory Group, four new concepts have been added to support the development of OWL axiom reference set:
- 733073007 |OWL axiom reference set|
- 733929006 |General concept axiom|
- 734147008 |OWL ontology header|
- 734146004 |OWL ontology namespace|
2.3.3 Dentistry Odontogram refset
- Odontogram - in anticipation of release the Odontogram Refset in August 2017, all supporting concepts were added following approval at the London meeting of the Dentistry project group
2.3.4 New attributes and values
Attribute (referencedComponentId_term) | rangeConstraint | Domain constraint |
|---|---|---|
| Before | << 71388002 |Procedure (procedure)| | Clinical finding Event |
| Component | << 123037004 |Body structure (body structure)| OR << 410607006 |Organism (organism)| OR << 105590001 |Substance (substance)| OR << 123038009 |Specimen (specimen)| OR << 260787004 |Physical object (physical object)| OR << 373873005 |Pharmaceutical / biologic product (product)| OR << 419891008 |Record artifact (record artifact)| | Clinical finding Event |
| Due to | << 404684003 |Clinical finding (finding)| OR << 272379006 |Event (event)| OR << 71388002 |Procedure (procedure)| | Clinical finding Event |
| During | << 71388002 |Procedure (procedure)| | Clinical finding Event |
| During AND/OR after | << 404684003 |Clinical finding (finding)| OR << 71388002 |Procedure (procedure)| | Clinical finding Event |
| Finding context | << 410514004 |Finding context value (qualifier value)| | Finding with explicit context (situation) |
| Finding informer | << 420158005 |Performer of method (person)| OR << 419358007 |Subject of record or other provider of history (person)| OR << 444018008 |Person with characteristic related to subject of record (person)| | Clinical finding |
| Has basis of strength substance | < 105590001 |Substance (substance)| | Pharmaceutical / biologic product (product) |
| Has disposition | < 726711005 |Disposition (disposition)| | Substance |
| Has interpretation | << 260245000 |Finding values (qualifier value)| OR << 263714004 |Colors (qualifier value)| | Clinical finding |
| Has presentation strength denominator unit | < 258666001 |Unit (qualifier value)| | Pharmaceutical / biologic product (product) |
| Has presentation strength denominator value | < 260299005 |Number (qualifier value)| | Pharmaceutical / biologic product (product) |
| Has presentation strength numerator unit | < 258666001 |Unit (qualifier value)| | Pharmaceutical / biologic product (product) |
| Has presentation strength numerator value | < 260299005 |Number (qualifier value)| | Pharmaceutical / biologic product (product) |
| Pathological process | 263680009 |Autoimmune (qualifier value)| OR << 441862004 |Infectious process (qualifier value)| OR << 472963003 |Hypersensitivity process (qualifier value)| OR << 308490002 |Pathological developmental process (qualifier value)| | Clinical finding |
| Temporally related to | << 404684003 |Clinical finding (finding)| OR << 71388002 |Procedure (procedure)| | Clinical finding Event |
 2.4 SNOMED CT derived products
2.4 SNOMED CT derived products
2.4.1 ICD-10 map
The SNOMED CT to the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (© World Health organisation 1994) 2016 Version map (SNOMED CT to ICD-10 Map) is included in the SNOMED CT International release as a Baseline. The SNOMED CT to ICD-10 Map was created to support the epidemiological, statistical and administrative reporting needs of SNOMED International member countries and WHO Collaborating Centres.
The SNOMED CT to ICD-10 Map is released in Release Format 2 (RF2) only. It is located in the file der2_iisssccRefset_ExtendedMapFull_INT_20170131.txt, which is in the Map folder under Refset, in each of the three RF2 Release Type folders.
The SNOMED CT to ICD-10 Map is released as Refset 447562003 |ICD-10 complex map reference set (foundation metadata concept)|.
2.4.1.1 Content development activity summary
The map is a directed set of relationships from SNOMED CT source concepts to ICD-10 target classification codes. The SNOMED CT source domains for the MAP are limited to subtypes of 404684003 |clinical finding|, 272379006 |event| and 243796009 |situation with explicit context|. The target classification codes are ICD-10 2016 release.
2.4.1.2 Mapped content for July 2017
The map provided for the July 2017 release has been updated, and now represents a complete map from SNOMED CT International release to ICD-10 2016 version.
- 2373 new concepts added
We would welcome feedback on any issues that users of the map may detect when using the map. Issues should be submitted via mapping@snomed.org
3 Technical notes
3.1 Known Issues
Known Issues are content or technical issues where the root cause is understood, and the resolution has been discussed and agreed but has yet to be implemented. This can be due to a number of reasons, from lack of capacity within the current editing cycle, to the risk of impact to the stability of SNOMED CT if the fix were to be deployed at that stage in the Product lifecycle.
For the Snomed CT July 2017 International edition, the following Known Issues were identified, and agreed to be resolved in the next editing cycle (to be published in July 2017):
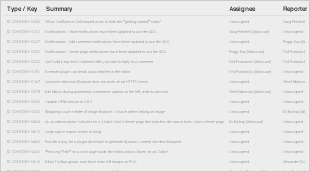
 3.2 Resolved Issues
3.2 Resolved Issues
Resolved issues are Known Issues which were not fixed as part of the previous release lifecycle, but which have now been resolved in the latest release - in this case the July 2017 International Edition. They can also be issues found during the Alpha and Beta testing of the current release, which were resolved before the final deployment of the associated Member release. Finally they can be issues which were reported or found during the testing phase, but which have been closed without any action taken.
The Resolved Issues for the Snomed CT July 2017 International edition can be found here:

 3.3 Technical updates
3.3 Technical updates
3.3.1 RF2 package naming convention
The naming convention of the RF2 package zip file has been improved, in order to bring it into line with the ISO8601 standard. A "Z" suffix has therefore been added to this package name (but not to internal file names), and will be added to all release package names going forward. This ensures that the time in the release package name is set in the correct context, namely UTC. In this case then, instead of the July 2017 International Edition release package being called:
SnomedCT_InternationalRF2_Production_20170731T120000.zip
It has instead been called:
SnomedCT_InternationalRF2_Production_20170731T120000Z.zip
3.3.2 Update to case significance assignment, in line with the RF2 specification
The incorrect assignment of case significance for descriptions has been fixed according to the RF2 specification. The issue was that the value 900000000000448009 |Entire term case insensitive| had not been used due to the data migration from RF1 and limitation of authoring tools. In fact, a majority of SNOMED CT descriptions should have been ‘Entire term case insensitive’. These terms can be freely switched to lower or upper case without impact to meaning. The technical batch changes have therefore been made to the case significance value to address this issue. For full details please see the document here: IHTSDO-950 Update of case significance assignment following the RF2 specification
3.3.3 Addition of the MRCM Refsets to the International Edition package
The MRCM Refsets, which were published in February 2017 as a Beta release, have now been included in the International Edition Release package. The following four new files have been incorporated into the Metadata folder, which is a sub-folder of the Refset folder in the Release package:
- der2_sssssssRefset_MRCMDomainDelta_INT_20170731.txt
- der2_cissccRefset_MRCMAttributeDomainDelta_INT_20170731.txt
- der2_ssccRefset_MRCMAttributeRangeDelta_INT_20170731.txt
- der2_cRefset_MRCMModuleScopeDelta_INT_20170731.txt
3.3.4 Improvements to the RefsetDescriptor file
The refsetDescriptor file has been improved in order to include descriptors for the pre-defined reference set types. It also now includes refsetDescriptor records to described the four new MRCM refsets now included in the International Edition from July 2017 onwards. Please see section 3.3.3. for further details of these refsets.
3.3.5 Addition of a new SEP refset to the International Edition package
This refset is being added in order both to assist with the interim solution for the Additional, non-defining Relationships, and also to enable us to further improve our Quality Assurance processes. Please see the following page for details of the proposed new Refset: SEP maps and refset
3.3.6 Removal of the SNOMED CT RT Identifier refset from the International Edition package
As has been communicated previously (across various forums) over the past several years, in 2009 SNOMED International's General Assembly decided that effective April 26, 2017 SNOMED International would no longer license the use of SNOMED antecedent (old) works. This decision received the clinical support of the World Association of Societies of Pathology and Laboratory Medicine, as they had identified the same risks. From April 26, 2017, therefore, SNOMED International is no longer maintaining antecedent versions of SNOMED, including SNOP, SNOMED, SNOMED II, SNOMED International (3.x), and SNOMED RT. Since both clinical science and terminology have evolved considerably since its development, the use of these older versions may involve risk to patients and safety issues.
SNOMED International cannot be responsible for this risk, and therefore in the original announcement strongly urged users of these antecedent versions to migrate to SNOMED CT as soon as possible.
As part of this process, the antecedent SNOMED Identifier reference set will be withdrawn and no longer be available in the International Edition release.
To provide a shared resource and a single source of truth on these antecedent issues we have assembled a set of FAQs available at our SNOMED International website here: https://ihtsdo.freshdesk.com/support/solutions/folders/4000013539
3.3.7 RF2 package format
For future reference, the RF2 package convention dictates that it contains all relevant files, regardless of whether or not there is content to be included in each particular release. Therefore, the package contains a mixture of files which contain both header rows and content data, and also files that are intentionally left blank (including only a header record). The reason that these files are not removed from the package is to draw a clear distinction between
- ...files that have been deprecated (and therefore removed from the package completely), due to the content no longer being relevant to RF2 in this or future releases, and
- ...files that just happen to contain no data in this particular release (and are therefore included in the package but left blank, with only a header record), but are still relevant to RF2, and could therefore potentially contain data in future releases.
This allows users to easily distinguish between files that have purposefully been removed or not, as otherwise if files in option 2 above were left out of the package it could be interpreted as an error, rather than an intentional lack of content in that release.
3.3.8 Early visibility of impending change in the January 2018 International edition
Please see the following page for details of all upcoming changes planned for January 2018 and beyond: January 2020 Early Visibility Release Notices - Planned changes to upcoming SNOMED International Release packages
NOTE: To access any of the links in the pdf document, please visit the Release Notes online here: https://confluence.ihtsdotools.org/display/RMT/SNOMED+CT+July+2017+International+Edition+-+SNOMED+International+Release+notes |
Approvals
|
Draft Amendment History
|
