For successful SNOMED CT adoption, it is important that the terminology conforms to the requirements of the specific use cases in the given country or within a given organization. In most situations this requires adjusting or extending the terminology, which can be done in various ways. SNOMED International authorizes Members and Affiliates to maintain and deliver additional components and derivatives known as SNOMED CT Extensions. These extensions are the formal way of conforming SNOMED CT to suit specific user requirements.
| Anchor |
|---|
| _Toc261806279 |
|---|
| _Toc261806279 |
|---|
|
| Anchor |
|---|
| _Toc268347160 |
|---|
| _Toc268347160 |
|---|
|
Extensions
Members may maintain and deliver additional terminology components and derivatives that adapt the terminology to meet specific national requirements. Moreover, SNOMED International Affiliates may also maintain and deliver additional terminology components and derivatives that adapt the terminology to meet the needs of a particular organization, customer or software solution. Extensions can therefore be distinguished as either National Extensions or Affiliate Extensions.
A National Extension may include:
- translation into the national language or adaption to a national dialect;
- additional content to support national policy objectives, a national drug dictionary or other specific requirements;
- derivatives that configure use of SNOMED CT content by specifying subsets of content to be used for particular purposes;
- derivatives that map other code systems used in that country to or from SNOMED CT.
An Affiliate Extension may include:
- additional content to enable a health provider organization or clinical specialty group to address its priority use cases;
- derivatives that configure use of SNOMED CT in ways that reflect the needs of a health provider organization or specialty;
- derivatives that configure the way SNOMED CT is used or presented to different customers using particular software applications;
- derivatives that map local or proprietary code systems to or from SNOMED CT.
Development of National or Affiliate Extensions requires the NRC or the Affiliate to have a Namespace Identifier, which is assigned by SNOMED International. A Namespace Identifier is used to identify the origin and the affiliation of each extension component. An NRC has the responsibility to ensure that any deployment of SNOMED CT and other SNOMED International terminology product conforms to SNOMED International standards. An NRC therefore serves as a point of contact for Affiliates to obtain guidance and advice in the development and maintenance of Affiliate Extensions.
| Anchor |
|---|
| _Toc261806280 |
|---|
| _Toc261806280 |
|---|
|
Extensions and Editions
The SNOMED CT International Edition can be used without any Extensions. However, a SNOMED CT Extension cannot be used on its own because all Extensions are dependent on the International Edition, and some Extensions are also dependent on other Extensions. Therefore, for each Extension there is a corresponding Edition that includes the Extension, the International Edition and any other Extensions on which it depends.
Figure 12 A National Edition includes both the National Extension and the International Edition
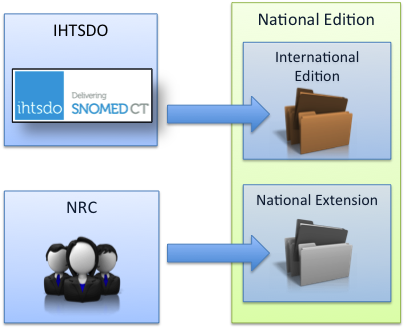 | All organizations that maintain Extensions should make their Extension available as a separate set of files, even if they also provide a pre-merged Edition. This allows validation of the constituent parts of the Edition and also supports merges to produce bespoke Editions that combine several Extensions. |
| Anchor |
|---|
| _Toc261806281 |
|---|
| _Toc261806281 |
|---|
|
| Anchor |
|---|
| _Toc268347161 |
|---|
| _Toc268347161 |
|---|
|
Translation
| Anchor |
|---|
| _Toc261806283 |
|---|
| _Toc261806283 |
|---|
|
The International Release of SNOMED CT contains terms in US English, GB English and Spanish. However, SNOMED CT is designed to support multilingual use by enabling terms in any language to be linked to the same concepts. Many of the current Members do not use English as the primary language in the delivery of healthcare, and therefore Members may require a translation of SNOMED CT to achieve a national terminology resource and to support users in the correct interpretation of the SNOMED CT content. The responsibility for translation is with those Members that use a particular language in the specific Member country. However, while Members are permitted and encouraged to translate SNOMED CT, this is not an obligation of membership. Each Member should consider whether or not to translate SNOMED CT into the native language and/or dialect, given the actual visions for use of SNOMED CT within that Member country.
A number of Members have already translated SNOMED CT into their native languages. These translations are managed within each of the particular Member countries as National Extensions and have been created based on SNOMED International regulations but according to the particular Member's requirements. Each translation may be shared with other Members, and all Members have an obligation according to the Articles of Association to share their translations upon request.
Translation approaches
Translating SNOMED CT does not necessarily mean a translation of the full SNOMED CT International Edition. An Member can decide to translate parts of SNOMED CT to obtain a core set of components to be used within the country. Another approach is to translate concepts that are to be applied within a specific organization, clinical domain, etc., which means a more use-case driven translation.
| Anchor |
|---|
| _Ref260990467 |
|---|
| _Ref260990467 |
|---|
|
Figure 13 Different levels of SNOMED CT translation 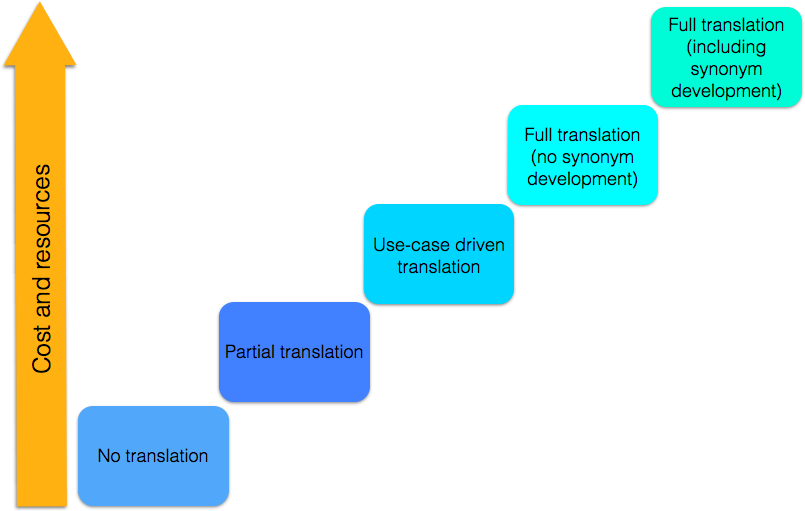
| Anchor |
|---|
| _Toc261806282 |
|---|
| _Toc261806282 |
|---|
|
| Anchor |
|---|
| _Toc261806285 |
|---|
| _Toc261806285 |
|---|
|
Translation Policy
SNOMED International believes that translations of SNOMED CT are very important because a translation can support SNOMED CT adoption due to enhanced understanding of the SNOMED CT content amongst the end users. Therefore SNOMED International has initiated a translation policy that reimburses a Member country for:
- 100% of the cost of translating concepts, which will be considered the "core" or starter set;
- The cost of translating one implementation document; and
- The cost of one SNOMED CT course.
(The maximum payment for each Member will be 150,000 USD.)
More information about SNOMED International Translation Policy can be found here: http://snomed.org/TranslationPolicy
Translation Guidance
Together with Members, SNOMED International has produced documentation that assists Members who decide to translate SNOMED CT. The documentation should support effective translation processes and enable Members to learn from the successes of those that have already set out on this task. The documentation can be found on the SNOMED International website and includes the following:
|
|---|
| This document describes important issues relevant to the translation of SNOMED CT, formulates recommendations, and establishes guidelines. The general guidelines may be used as a template to develop national guidelines. In addition to linguistic and terminological guidelines, the document includes general recommendations about the steps involved in a translation workflow process and information about source documents or references that must be made available to those involved in the translation process. |
|
|---|
| This document is a companion guideline to be used with the "Guidelines for the Translation of SNOMED CT" and offers recommendations on management of a translation project. The purpose is to help new Members undertaking translations to avoid mistakes and enable them to build on existing experiences. |
|
|---|
| This document describes the use of SNOMED International methodology and toolkit for evaluating SNOMED CT translation quality and has been developed to conform to SNOMED International Quality Assurance Framework. The document also includes the actual toolkit and therefore is useful for any Members looking for translation support materials. |
|
|---|
SNOMED International Quality Assurance Framework, with its supporting SNOMED International Quality Assurance Toolkit, was developed by SNOMED International Quality Assurance Committee and aims to support the development and reporting of Quality Metrics across all areas of SNOMED International activity, including organizational processes and support, data products (e.g. terminology reference data, mappings, translations, subsets), documentation, and SNOMED International-responsible services and tooling provisions.
Other resources are also available, mostly concerning the practical experiences of those who have undertaken translation development. |
| Anchor |
|---|
| _Toc261806286 |
|---|
| _Toc261806286 |
|---|
|
| Anchor |
|---|
| _Toc268347162 |
|---|
| _Toc268347162 |
|---|
|
Reference set development
Many implementations of SNOMED CT do not require users to have access to the full set of content. Often just a part of the data related to a particular discipline or organization will be needed. It is possible to constrain the content required by the use of the subsets or Reference sets.
Reference sets are sets of references to SNOMED CT components. Simple reference sets contain just the information needed to define a subset. However Reference sets can be richer than subsets, as they can also include further information about the components and have been defined for other purposes such as mappings to other vocabularies or providing alternative hierarchical structures.
Reference sets are distributed as tab-delimited text files with each row including a SNOMED CT component identifier for the Reference set member. There are also several standard fields that support versioning and additional fields defined to meet the requirements of specific Reference set types.
Full details on Reference sets and the Reference set format may be found in the Reference Sets Guide, which is part of the Technical Implementation Guide: http://snomed.org/tig.